
Introduction
Creating effective skincare products that deliver visible results without harmful ingredients is more complex than most entrepreneurs realize. A single formulation error—whether it's an incompatible preservative system, unstable emulsion, or inadequate safety testing—can result in product recalls, customer complaints, and significant financial losses.
The stakes are particularly high now, as 93% of U.S. adults have used "clean" personal care products in the past year, creating unprecedented demand for toxin-free formulations.
This consumer shift toward natural skincare has reshaped product development requirements. Brands must now navigate stricter preservation challenges, stability testing protocols, and evolving regulatory standards while delivering products that perform as well as conventional alternatives.
This article provides expert insights into formulation fundamentals, development processes, testing requirements, and how to successfully navigate the formulation landscape as an entrepreneur or brand owner.
TLDR: Key Takeaways
- Skincare formulation management requires specialized expertise in chemistry, safety testing, and regulatory compliance to create stable, effective products
- Natural formulations require "hurdle technology" to overcome preservation and stability challenges
- Physical, chemical, and microbiological stability testing is non-negotiable for product safety
- The Modernization of Cosmetics Regulation Act (MoCRA) now mandates facility registration and safety substantiation for all cosmetic brands
- FDA cGMP-compliant manufacturers reduce development risks with proven toxin-free formulations
What Is Skincare Formulation Management?
Skincare formulation management is the systematic process of developing, testing, and maintaining product formulas that are safe, effective, and stable throughout their intended shelf life.
It extends far beyond simply mixing ingredients in a bowl—it's a technical discipline that requires chemistry expertise, regulatory knowledge, and rigorous quality control processes.
Beyond Basic Formulation
The difference between basic formulation and comprehensive formulation management is significant. Basic formulation involves creating a product that works initially, while formulation management includes:
- Ongoing stability monitoring to ensure products maintain their integrity over time
- Reformulation processes when ingredient suppliers change or raw materials vary
- Batch consistency systems that guarantee every production run matches the original specification
- Performance validation through both laboratory testing and real-world use scenarios
Key Components of Formulation Management
Effective formulation management integrates several critical elements:
- Identifying research-backed actives, compatible carriers, and effective preservation systems that align with product goals and safety standards
- Determining precise percentages of each ingredient to maximize efficacy while maintaining stability and sensory appeal
- Developing preservation methods that protect products from contamination without relying on synthetic preservatives consumers avoid
- Creating sensory experiences (absorption rate, skin feel, fragrance) that meet consumer expectations while maintaining product integrity
- Conducting testing to verify that products deliver their promised benefits under real-world conditions
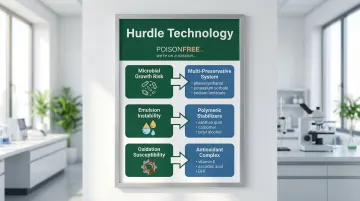
Natural Formulation Challenges
Formulating with natural and toxin-free ingredients presents distinct challenges compared to conventional cosmetics. Natural ingredients often serve as nutrient-rich media that favor microbial growth, requiring more sophisticated preservation strategies.
Plant-based emulsifiers can be more sensitive to temperature fluctuations and pH changes, potentially leading to phase separation or texture changes during storage. Natural oils and botanical extracts are also prone to oxidation, which can cause rancidity, color shifts, and odor changes over time.
Formulators address these challenges by implementing robust antioxidant systems and carefully controlling processing parameters to ensure product stability.
The Cosmetic Chemist's Role
Qualified cosmetic chemists translate product concepts into workable formulas. They typically hold degrees in chemistry, biology, or related sciences and gain specialized formulation expertise through programs like the Society of Cosmetic Chemists Certificate Program.
Their responsibilities extend beyond ingredient selection to include regulatory review, scale-up planning, and troubleshooting production issues that arise when moving from laboratory batches to commercial manufacturing.
The Formulation Development Process: From Concept to Production
Stage 1: Concept Definition and Target Product Profile
Every successful formulation begins with a clear product concept that defines:
- Target skin types and concerns
- Desired benefits and performance claims
- Texture preferences and sensory expectations
- Natural versus synthetic ingredient preferences
- Price point and market positioning
This target product profile serves as the roadmap for all subsequent development decisions.
Stage 2: Ingredient Research and Selection
The ingredient selection process focuses on several critical factors:
- Identifying actives with clinical evidence supporting their claimed benefits
- Ensuring ingredients work synergistically without chemical interactions that compromise stability
- Finding effective antimicrobial systems that meet clean beauty standards
- Confirming ingredient quality, organic certifications, and accurate INCI naming
For toxin-free formulations, this stage requires identifying plant-based alternatives to synthetic ingredients while maintaining comparable performance. Manufacturers with large inventories of vetted raw materials can significantly reduce sourcing delays during this phase.
Stage 3: Initial Formulation and Bench Testing
Prototype development involves multiple iterative steps:
- Create small-batch samples with varying ingredient ratios
- Evaluate texture, absorption rate, and sensory experience
- Adjust pH levels to optimize ingredient stability and skin compatibility
- Test different emulsification systems for stability
- Refine formulas based on initial performance observations
This iterative process typically produces multiple prototype versions before arriving at a candidate formula for formal testing.
Stage 4: Stability and Safety Testing Protocols
Comprehensive testing validates that formulations remain safe and effective throughout their shelf life:
- Storing products at elevated temperatures (typically 40°C at 75% relative humidity) for 3-6 months to predict 2-3 year shelf life under normal conditions
- Following ISO 11930 protocols by introducing specific microorganisms and measuring reduction in microbial count over 28 days to verify antimicrobial effectiveness
- Tracking pH stability over time, as shifts can indicate ingredient degradation or chemical reactions that compromise product safety
- Subjecting products to temperature extremes to test emulsion stability during shipping and storage
Stage 5: Scale-Up Considerations
Transitioning from laboratory beakers to production tanks introduces several challenges:
- Natural emulsions often behave differently under high-shear production mixing compared to lab stirring
- Production equipment requires validated temperature control protocols for heating and cooling rates
- Natural raw materials can vary by harvest, requiring strict raw material specifications
- Formulas must work with available filling and packaging equipment
Pilot batches at intermediate scales help identify and resolve these issues before full production runs.

Development Timelines
Development timelines vary based on formulation complexity:
- Private label products: 3-6 months from concept to market
- Custom formulations: 12-20 weeks for standard products
- Complex formulations: 8-20+ weeks for products with advanced actives or specific claims
Working with manufacturers who maintain large inventories of raw materials and have proven formulation libraries can significantly accelerate development by eliminating ingredient sourcing delays.
Key Ingredients in Natural Skincare Formulations
Primary Functional Categories
Natural skincare formulations rely on four main ingredient categories:
Humectants attract and retain moisture in the skin. Common natural options include glycerin, sodium hyaluronate (hyaluronic acid), and honey.
Overuse can create sticky textures that consumers find unpleasant, so balancing hydration with sensory appeal is essential.
Emollients soften and smooth skin while providing sensory appeal. Plant oils (jojoba, argan, rosehip), butters (shea, cocoa), and plant-derived triglycerides create the luxurious feel consumers expect.
Occlusives form a protective seal to prevent water loss. Natural options include beeswax, heavier plant oils, and plant waxes—particularly important in barrier repair and intensive moisturizing products.
Active ingredients deliver targeted benefits: antioxidant protection (vitamin C, vitamin E), cell renewal support (retinol alternatives), or soothing properties (chamomile, calendula, green tea).
Natural Versus Synthetic Preservatives
Preservation represents one of the most significant formulation challenges in natural skincare. Traditional synthetic preservatives like parabens and formaldehyde donors offer broad-spectrum protection across wide pH ranges.
Natural alternatives—including benzyl alcohol, salicylic acid, and sorbic acid—typically have narrower effectiveness ranges and work only at specific pH levels.
Key preservation challenges include:
- Shorter shelf life: Natural systems typically last 12-18 months versus 24-36 months for synthetic options (unless combining multiple preservation methods)
- pH restrictions: Organic acids work only at pH levels below 6.0, limiting formulation flexibility
- Higher concentrations: Natural preservatives often require more material, which can affect texture and feel
Sourcing Quality Toxin-Free Ingredients
Successful natural formulation depends on ingredient quality and verification:
- Supplier verification: Establishing relationships with reputable suppliers who provide certificates of analysis and maintain consistent quality standards
- Organic certifications: Understanding standards like COSMOS and USDA Organic, which have strict criteria for natural and organic claims
- INCI name accuracy: Ensuring ingredient labels use proper International Nomenclature of Cosmetic Ingredients (INCI) naming for regulatory compliance
Working with experienced manufacturers who've spent decades building supplier networks and quality protocols can significantly reduce ingredient-related risks. For example, companies with 30+ years formulating toxin-free products have already vetted suppliers and established verification systems that new brands can leverage.

Stability Testing and Quality Control
Three Critical Types of Stability Testing
Comprehensive stability testing evaluates three distinct aspects of product integrity:
Physical Stability Testing tracks visible changes including:
- Phase separation in emulsions
- Color shifts indicating ingredient degradation or chemical reactions
- Texture changes (viscosity increases or decreases)
- Precipitation or crystallization of ingredients
Chemical Stability Testing measures:
- pH shifts that indicate ingredient breakdown
- Active ingredient degradation through analytical testing
- Oxidation of oils and botanical extracts
- Interaction between ingredients that compromise efficacy
Microbiological Stability Testing verifies:
- Preservative system effectiveness through challenge testing
- Absence of contamination in finished products
- Maintenance of antimicrobial protection throughout shelf life
Accelerated Stability Testing Methods
Accelerated aging protocols allow formulators to predict real-world shelf life without waiting years for results.
By storing products at elevated temperatures (typically 40°C) and high humidity (75% RH) for 3-6 months, formulators can simulate 2-3 years of normal storage conditions.
These tests reveal potential stability issues including:
- Emulsion breaking or phase separation
- Active ingredient degradation rates
- Preservative system failures
- Packaging compatibility problems
Batch Consistency Protocols
Quality control extends beyond initial formulation to ensure every production batch meets specifications. FDA cGMP-compliant facilities like Poison-Free Private Label's Northern Texas manufacturing operation implement systematic checks at multiple stages:
- Raw material testing: Verifying ingredient identity and purity before use
- In-process controls: Monitoring pH, viscosity, and other parameters during manufacturing
- Finished product testing: Confirming final products meet all specifications
- Retention samples: Maintaining samples from each batch for ongoing stability monitoring
Packaging Compatibility Testing
Packaging actively affects product stability, not just containing it. Test for these compatibility factors:
- Chemical interactions: Whether ingredients migrate into packaging (sorption) or packaging chemicals transfer into products
- Physical stress: Testing for container cracking, pump failure, or closure issues
- Barrier properties: Ensuring packaging protects light-sensitive or oxygen-sensitive ingredients
- Stability impact: How different packaging materials affect product shelf life
Recent regulatory guidelines from agencies like China's NIFDC explicitly require packaging compatibility testing reports, influencing global best practices and raising quality standards across the industry.

Industry Failure Rates
Inadequate stability testing carries significant financial risks. Industry data indicates that formulation instability and microbial contamination drive most product failures, with preservative system inadequacy being particularly common in natural formulations.
The cost implications extend beyond lost product inventory to include customer refunds, brand reputation damage, and potential liability issues if products cause adverse reactions.
Regulatory Compliance and Safety Standards
FDA Regulations and cGMP Requirements
The Modernization of Cosmetics Regulation Act of 2022 (MoCRA) fundamentally changed U.S. cosmetic compliance. This legislation introduced four major requirements for all cosmetic brands:
Facility Registration & Product Listing
Manufacturers must register facilities with the FDA biennially. Responsible persons must list each marketed product annually with complete ingredient information, creating a comprehensive regulatory database.
Safety Documentation Requirements
Brands must maintain records proving product safety. While the FDA doesn't mandate specific tests, your documentation must be scientifically robust and demonstrate adequate safety substantiation.
Good Manufacturing Practices (cGMP)
FDA cGMP compliance ensures manufacturing processes meet strict quality standards. This covers facility sanitation, personnel training, equipment maintenance, and production controls. FDA cGMP-compliant manufacturers like Poison-Free Private Label maintain these standards to ensure consistent product quality and regulatory adherence.
Labeling Requirements and Ingredient Disclosure
FDA regulations mandate specific labeling practices:
- Ingredients must be listed in descending order using proper INCI names
- Components at 1% or less can be listed in any order after higher-concentration ingredients
- Fragrance and flavor components can be listed collectively without disclosing individual elements
- Color additives require specific identification using approved names
Claims Substantiation
Marketing language must be substantiated with appropriate evidence. Claims about product benefits, efficacy, or performance require supporting data from clinical studies, consumer perception studies, or laboratory testing.
Drug claims like "treats acne" or "SPF protection" trigger OTC drug regulations requiring FDA monograph compliance.
Safety Assessments and Documentation
While cosmetics don't require FDA pre-market approval, maintaining comprehensive safety documentation is essential for:
- Liability protection: Demonstrating due diligence in ensuring product safety
- Consumer confidence: Providing transparency about safety testing and ingredient selection
- Regulatory compliance: Meeting MoCRA safety substantiation requirements
- Recall preparedness: Having documentation ready if safety questions arise
The FDA now has authority to mandate recalls if products are suspected of causing serious adverse health consequences, making proactive safety assessment more critical than ever.

Choosing the Right Formulation Partner
Key Selection Criteria
Selecting the right manufacturing partner is one of the most important decisions for skincare entrepreneurs. Critical evaluation criteria include:
- Experience with natural formulations — Manufacturers specializing in toxin-free products understand the unique challenges of natural preservation systems, plant-based ingredient variability, and clean beauty consumer expectations
- FDA cGMP compliance — cGMP-compliant facilities ensure your products meet regulatory quality standards and reduce liability risks
- Minimum order quantities — Industry standard MOQs typically range from 1,000-10,000 units, while entrepreneur-friendly manufacturers offering MOQs as low as 500 units enable market testing without large capital commitments
- Development turnaround times — Realistic timelines from concept to production help with business planning and launch scheduling
- Existing formula library — Manufacturers with extensive proven formulations can significantly accelerate time-to-market through private label or customized stock formulation options
Advantages of Low MOQ Manufacturing
One criterion deserves special attention for new brands: minimum order quantities.
For entrepreneurs testing market viability, low MOQs offer several strategic advantages:
- Cuts initial capital investment by 50-80% compared to standard MOQs
- Test multiple product variations without overcommitting
- Reduces inventory risk for unproven brands
- Pivot quickly based on market feedback
- Launch faster without large production commitments
Experience as a Risk Reducer
Experienced manufacturers bring substantial value to new brands. Look for partners with decades of formulation experience and extensive libraries of proven formulations. This expertise translates to:
- Established ingredient sourcing networks ensuring quality and consistency
- Proven preservation systems that pass challenge testing
- Knowledge of common formulation pitfalls and how to avoid them
- Faster development timelines through existing formula modification rather than starting from scratch
- Regulatory expertise in labeling, claims verification, and compliance documentation
This expertise significantly reduces the technical and financial risks associated with bringing new skincare products to market.
Common Formulation Challenges and Solutions
Challenge #1: Achieving Stability with Natural Ingredients
Natural ingredients present inherent stability challenges that synthetic alternatives don't face.
The stability challenge: Natural emulsifiers lack the robust stabilizing power of ethoxylated synthetic emulsifiers and are sensitive to pH changes and electrolytes.
Plant oils oxidize over time, causing rancidity and color changes. Botanical extracts can vary in potency and composition between harvests.
Solutions:
- HLB balancing: Carefully selecting emulsifiers based on Hydrophile-Lipophile Balance values and pairing high and low HLB emulsifiers creates more stable interfaces
- Polymeric stabilizers: Adding natural thickeners like xanthan gum improves viscosity and emulsion stability
- Antioxidant systems: Combining tocopherol (vitamin E), rosemary extract, and ascorbyl palmitate provides synergistic protection against oxidation
- pH optimization: Maintaining optimal pH ranges (typically 5.0-6.0) maximizes preservative efficacy and ingredient stability
Challenge #2: Balancing Efficacy with Clean Ingredient Standards
Consumers expect natural products to perform as well as conventional alternatives while meeting strict clean beauty standards.
The efficacy gap: Many highly effective synthetic actives don't have natural equivalents with identical performance profiles.
Natural alternatives may require higher concentrations or longer use periods to deliver comparable results.
Solutions:
- Research-backed natural actives: Identifying botanical extracts and natural compounds with clinical evidence supporting their efficacy
- Synergistic combinations: Using multiple natural actives that work together to achieve results comparable to single synthetic ingredients
- Delivery system optimization: Enhancing penetration and bioavailability of natural actives through appropriate carrier systems
- Realistic expectations: Educating consumers about appropriate timelines for seeing results with natural formulations
This expertise comes with experience. Formulators with deep natural ingredient knowledge—such as those who've developed 100+ toxin-free formulations over decades—can identify effective alternatives that maintain product performance while meeting clean beauty standards.
Challenge #3: Managing Production Costs While Maintaining Quality
Natural and toxin-free formulations often involve higher ingredient costs than conventional products.
The cost challenge: Premium natural ingredients, rigorous testing requirements, and smaller production batches can significantly increase per-unit costs. This can potentially price products out of competitive ranges.
Solutions:
- Strategic ingredient sourcing: Building relationships with reliable suppliers who offer competitive pricing for quality ingredients
- Optimized batch sizing: Finding the sweet spot between per-unit cost efficiency and inventory management
- Formula efficiency: Designing formulations that achieve desired results with cost-effective ingredient combinations
- Value communication: Educating consumers about the quality and safety benefits that justify premium pricing
Manufacturers specializing in toxin-free formulations—like Poison-Free Private Label with its large inventory of raw materials and 30 years of supplier relationships—often have established networks and economies of scale that allow competitive pricing even for smaller batch orders.
Frequently Asked Questions
What is skincare formulation?
Skincare formulation is the scientific process of combining ingredients in specific ratios to create safe, stable, and effective products. It requires expertise in chemistry, ingredient interactions, and preservation systems to ensure safety throughout shelf life.
How long does it typically take to develop a new skincare formulation?
Development timelines vary from 3-6 months for simple formulas using existing bases to 12+ months for complex custom products requiring extensive testing. The timeline depends on complexity, ingredient availability, and whether you're using private label versus fully custom development.
What's the difference between natural and synthetic skincare ingredients in formulation?
Natural ingredients are plant or mineral-derived and require different preservation approaches compared to lab-synthesized compounds. They're more prone to oxidation, microbial growth, and batch variability, requiring sophisticated formulation strategies to ensure stability.
What are the minimum order quantities for custom formulation with most manufacturers?
Industry standard MOQs typically range from 1,000-10,000 units depending on the manufacturer and product complexity. Poison-Free Private Label offers MOQs as low as 500 units, enabling new brands to test market viability without large capital commitments or excessive inventory risk.
Do I need FDA approval to sell skincare products?
No, cosmetics don't require pre-market FDA approval. However, manufacturers must follow cGMP guidelines, facilities must register with the FDA under MoCRA, products must be listed annually, and brands must maintain safety substantiation documentation. Products must be safe and properly labeled according to FDA regulations to avoid enforcement actions.
How do I know if a manufacturer's formulations are truly toxin-free?
Look for manufacturers with FDA cGMP compliance and established track records—Poison-Free Private Label has 30 years in business with transparent ingredient sourcing. Ask about ingredient verification processes, supplier certifications, and testing protocols. Reputable manufacturers should provide complete INCI ingredient lists and discuss quality control measures openly.


