
Introduction
Private label manufacturing offers a strategic entry point into the skincare market—allowing you to launch products without building costly infrastructure or developing formulations from scratch. According to industry analysis, the U.S. skincare market reached $22.90 billion in 2023 and continues growing at 4.2% annually, creating accessible opportunities for entrepreneurs and established brands.
Choosing the right manufacturer involves navigating critical pain points: minimum order quantities that can strain startup budgets, formulation quality that directly impacts brand reputation, compliance standards that vary widely across facilities, and turnaround times that affect your speed to market.
The wrong partner can mean excess inventory, regulatory headaches, or products that don't align with your brand values.
This guide profiles five leading U.S. private label skincare manufacturers across different specializations and price points, helping you identify the right partner for your business stage and brand vision.
TLDR
- Private label manufacturing cuts time-to-market by leveraging existing formulations instead of costly custom development
- FDA-compliant facilities offer MOQs from 500-5,000 units based on customization needs
- Prioritize cGMP certification, formulation library depth, and brand positioning alignment
- Stock formulations ship in 4-8 weeks versus 12-24 weeks for custom development
- Choose manufacturers matching your business stage to avoid cash flow strain
Overview of Private Label Skincare Manufacturing in the USA
Starting a skincare brand no longer requires a six-figure investment in manufacturing facilities. Private label manufacturing lets you select existing formulations or customize them, then package products under your own brand name—slashing capital requirements and getting you to market faster than building from scratch.
Market Landscape
The U.S. personal care contract manufacturing market reached $3.33 billion in 2024, with skincare products commanding 35.5% of revenue—the largest category share.
This segment is expanding at 7.9% annually through 2030, outpacing the broader skincare market's 4.2% growth rate.
Consumer demand is shifting toward clean, sustainable formulations that deliver proven results. Facial skincare ranks as the top category where consumers prioritize "clean" attributes.
Efficacy has become equally important—only 14% of U.S. buyers believe higher prices indicate better quality.
These market dynamics make choosing the right manufacturing partner critical. Domestic manufacturers offer distinct advantages that matter for both quality and speed.
Why Manufacture in the USA
Regulatory Assurance: The FDA's Modernization of Cosmetics Regulation Act of 2022 (MoCRA) requires Good Manufacturing Practices and facility registration for all cosmetic manufacturers. U.S. facilities must comply with ISO 22716 standards, ensuring consistent quality and safety.
Supply Chain Benefits: Domestic production eliminates international shipping delays, reduces tariff exposure, and enables better quality control through proximity.
Turnaround times for stock formulations typically run 4-8 weeks versus 12+ weeks for overseas manufacturing.
Communication & Oversight: Working with U.S. manufacturers allows for easier facility visits, real-time communication during production, and faster resolution of any formulation or packaging issues.
Top Private Label Skincare Manufacturers in the USA
These manufacturers were selected based on key criteria important to new brand owners:
- Compliance standards and certifications
- Formulation diversity and customization options
- MOQ flexibility for different business stages
- Production capacity and turnaround times
- Verified customer feedback and track record
Each represents different strengths to match your specific brand needs.
Cosmetic Solutions
Based in Boca Raton, Florida, Cosmetic Solutions operates a 100,000-square-foot Innovation Campus and has served emerging brands, established companies, and medical professionals since 1991.
Their focus centers on high-performance, science-driven formulations for anti-aging, clinical skincare, and spa-grade products.
What Differentiates Them: Extensive R&D capabilities combined with turnkey services including custom formulation, premium packaging, and regulatory support. Their ISO 22716 certification and FDA registration show commitment to quality standards, while their formulation library spans multiple clinical and medical-grade categories.
| Category | Details |
|---|---|
| Minimum Order Quantity (MOQ) | 100-500 units for private label stock formulations; higher for custom development |
| Certifications & Compliance | FDA-registered, ISO 22716 certified, cGMP compliant |
| Specialization & Key Services | Anti-aging and clinical skincare formulations; full-service R&D; custom formulation; premium packaging design; regulatory guidance |
RainShadow Labs
Operating from St. Helens, Oregon since 1983, RainShadow Labs brings over 40 years of experience as a full-service personal care manufacturer specializing in clean and natural formulations. They prioritize botanical ingredients and sustainable practices for eco-conscious brands.
What Differentiates Them: Maintains hundreds of market-ready stock formulas using natural ingredients, making them ideal for brands positioning around clean beauty. Their cruelty-free commitment and organic capabilities attract brands seeking certifications that resonate with environmentally aware consumers.
| Category | Details |
|---|---|
| Minimum Order Quantity (MOQ) | Low MOQs on bulk and filling; stock formulas available for private label customers |
| Certifications & Compliance | FDA registered, ISO 22716:2007 certified, cGMP certified, Cruelty-Free |
| Specialization & Key Services | Clean & natural formulations with botanical ingredients; hundreds of stock formulas; custom manufacturing; sustainability-focused practices |
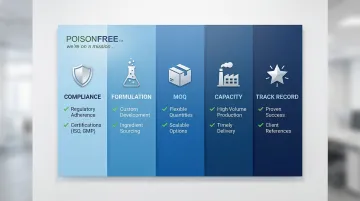
Poison-Free Private Label
A family-owned manufacturer based in Northern Texas with 30 years of experience, founded by Sindi Holmlund who started formulating products in her kitchen to replace conventional items containing toxins. The company specializes in 100% toxin-free, petrochemical-free formulations made exclusively from plants, minerals, and vitamins.
What Differentiates Them: Entrepreneur-friendly policies designed for startups testing market fit:
- 500-unit MOQ (significantly lower than the 1,000-5,000 industry standard)
- All orders priced as if you bought 10,000 units—no small batch penalty
- Fast turnaround with orders typically shipping within a week
- Founder's philosophy: "if I won't use it, I won't make it and sell it to anyone else"
| Category | Details |
|---|---|
| Minimum Order Quantity (MOQ) | 500 units (significantly lower than industry standard) |
| Certifications & Compliance | FDA cGMP compliant facility |
| Specialization & Key Services | Over 100 toxin-free formulations using only plants, minerals, and vitamins; custom formulation services; large raw materials inventory for stock and custom products; fast turnaround (typically within a week); ideal for startups and entrepreneurs testing market fit |
Prime Matter Labs
For brands ready to scale, Prime Matter Labs in Miami Gardens, Florida offers high-volume production capabilities. Headquartered in Miami Gardens, Florida, Prime Matter Labs is a large-scale manufacturer known for high-performance formulations and scalability. They serve established brands, retailers, and companies with international distribution needs.
What Differentiates Them: Excels at moderate to high-volume production with automation technology that ensures consistency and efficiency. Their capabilities include comprehensive supply chain management and quality control systems designed for brands scaling beyond startup phase.
| Category | Details |
|---|---|
| Minimum Order Quantity (MOQ) | Typically 1,000+ units, geared toward moderate to large production runs |
| Certifications & Compliance | FDA-registered, cGMP compliant |
| Specialization & Key Services | High-volume production of moisturizers, serums, cleansers, and creams; full-service product development; supply chain management; quality control systems for scaling brands |
Malibu Labs
Based in Gardena, California, Malibu Labs specializes in clean, plant-based, and organic beauty formulas with over 20 years of experience. They've built a library of 1,500+ proprietary formulas and can produce up to 60,000 units per month.
What Differentiates Them: Offers comprehensive turnkey services including custom formulation, branding, marketing support, and manufacturing—all under one roof. Their focus on non-toxic, organic, and plant-based products for skin, hair, and body care makes them ideal for brands seeking full-service support with clean beauty positioning.
| Category | Details |
|---|---|
| Minimum Order Quantity (MOQ) | Flexible production capabilities; can produce up to 60,000 units per month |
| Certifications & Compliance | FDA-registered facility, MoCRA compliant, ISO 22716 standards |
| Specialization & Key Services | 1,500+ proprietary formulas; comprehensive turnkey services including custom formulation, branding, marketing, and manufacturing; focus on non-toxic, organic, and plant-based products |
How to Choose the Right Private Label Skincare Manufacturer
Common Mistakes Brands Make
Many entrepreneurs choose manufacturers based solely on price without evaluating quality, leading to reformulation costs or customer complaints later. Others ignore MOQ alignment with their business stage and cash flow, resulting in excess inventory that ties up capital.
Overlooking compliance and certification requirements can cause regulatory issues, while failing to assess formulation flexibility limits your ability to differentiate products.
Critical Evaluation Factors
Check compliance documentation first:
- FDA registration and cGMP adherence, specifically ISO 22716 compliance
- Certificates of Analysis (COA) and Safety Data Sheets (SDS) for quality control verification
- Incoming raw material testing and in-process controls (pH, viscosity)
- Finished product testing for microbiological safety and stability

Evaluate formulation capabilities:
- Depth of formulation library and customization options
- Ability to modify existing formulations to meet your specific needs
- In-house stability testing and microbiological testing
Assess communication and transparency. Responsive communication during evaluation typically indicates how they'll perform during production. Manufacturers who provide detailed answers about raw material sourcing, audit history, and production processes demonstrate trustworthiness.
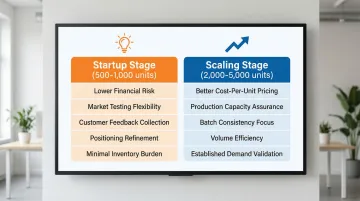
MOQ Considerations for Different Business Stages
Startups & First-Time Brands: Seek manufacturers offering 500-1,000 unit MOQs to test market demand without excessive inventory risk. Lower MOQs allow you to validate product-market fit, gather customer feedback, and refine positioning before committing to larger production runs.
Manufacturers like Poison-Free Private Label offer 500-unit minimums—ideal for entrepreneurs testing new concepts without tying up capital in excess inventory.
Established Brands Scaling Up: Once you've validated demand, prioritize production capacity, batch consistency, and cost-per-unit efficiency. Manufacturers with higher MOQs (2,000-5,000 units) typically offer better pricing and can handle growing order volumes without capacity constraints.
Alignment with Brand Values
If positioning as clean beauty, verify the manufacturer sources quality ingredients and maintains clear exclusion lists for controversial chemicals (parabens, sulfates, phthalates, synthetic fragrances).
Request full ingredient lists and review their formulation philosophy.
For sustainability-focused brands, confirm eco-friendly packaging options, ethical sourcing practices, and any relevant certifications (USDA Organic, Leaping Bunny). Request samples before committing to ensure formulation quality matches your standards and brand promises.
Key Trends Shaping Private Label Skincare Manufacturing in 2025
Clean Beauty Movement
Consumer demand for toxin-free, transparent formulations continues accelerating, with facial skincare ranking as the top category where buyers prioritize "clean" attributes.
However, today's consumers are redefining "clean" to include both safety and efficacy—products must deliver proven results, not just avoid controversial ingredients. Manufacturers are responding by reformulating products without parabens, sulfates, phthalates, and synthetic fragrances while maintaining performance standards.
This shift requires advanced formulation knowledge to replace traditional preservatives and stabilizers with effective natural alternatives.
Personalization and Customization
As consumers demand cleaner formulations, they also want products tailored to their unique needs. This shift toward personalized skincare solutions is driving manufacturers to offer flexible custom formulation services and smaller batch capabilities.
Leading facilities now support:
- Offering customizable formulations within their existing libraries
- Reducing MOQs for custom development projects
- Providing modular packaging options for personalized branding
- Supporting limited-edition and seasonal product launches

Sustainability and Ethical Sourcing
Consumer preference for eco-friendly packaging, carbon-neutral manufacturing, and ethically sourced ingredients is transforming manufacturer operations.
Leading facilities are adopting sustainable practices including renewable energy, water-saving technologies, and green logistics to meet market demand and regulatory pressures.
Certifications like Leaping Bunny (cruelty-free), USDA Organic, and Non-GMO Project Verified are becoming key differentiators. Brands should verify manufacturers can provide documentation supporting sustainability claims to avoid greenwashing accusations.
Conclusion
Choosing a private label manufacturer requires matching capabilities to your brand vision, business stage, and budget—not simply selecting the biggest or most well-known option.
The right partner provides appropriate MOQ flexibility, aligns with your positioning (clean beauty, clinical, high-volume), and demonstrates transparent communication about formulations and compliance.
Before committing to a manufacturer:
- Request samples from multiple manufacturers to evaluate quality firsthand
- Review their certifications thoroughly (FDA cGMP, organic certifications)
- Discuss formulation capabilities, MOQs, and turnaround times in detail
- Verify they provide Certificates of Analysis for every batch
The manufacturers covered in this guide offer diverse strengths, from high-volume production to niche clean beauty formulations. For entrepreneurs and startups specifically seeking toxin-free products with 500-unit MOQs and fast turnaround, Poison-Free Private Label offers 100+ clean formulations with pricing scaled to support new brands testing the market.
Frequently Asked Questions
How much does it cost to formulate a skincare product?
Stock formulations typically cost $5,000-$15,000 total for initial production, while custom development ranges from $5,000-$25,000 just for formulation fees before production. Private label models using existing formulations significantly reduce upfront costs.
Who formulates skincare?
Cosmetic chemists and formulators who specialize in ingredient interactions, stability testing, and regulatory compliance create skincare products. In private label manufacturing, manufacturers employ these experts and provide ready-made formulations, eliminating the need to hire your own chemist.
What is the 1% rule in skincare?
The 1% rule refers to FDA regulation 21 CFR 701.3, which requires ingredients above 1% concentration to be listed in descending order. Ingredients at 1% or less can be listed in any order after those above 1%.
What is the minimum order quantity for private label skincare?
MOQs typically range from 500 to 5,000 units depending on the manufacturer and formulation type. Startups should seek manufacturers with lower MOQs (500-1,000 units) to minimize risk, while established brands can negotiate better pricing with higher volumes.
How long does it take to manufacture private label skincare?
Production timelines vary from 4-8 weeks for stock formulations with materials in inventory to 12-24 weeks for custom formulations requiring ingredient sourcing, stability testing, and packaging setup. Factors like packaging complexity, order size, and current production schedules also impact turnaround time, so discuss specific timelines with your manufacturer early in the planning process.
What certifications should a skincare manufacturer have?
At minimum, manufacturers should be FDA registered and follow cGMP (Good Manufacturing Practices), ideally with ISO 22716 certification. Additional certifications like USDA Organic, Leaping Bunny (cruelty-free), or Non-GMO Project Verified depend on your brand positioning and target market.


