
Introduction
The U.S. skincare industry is booming. With the market valued at $22.90 billion in 2023 and growing at 4.2% annually, entrepreneurs and small brands struggle with a costly barrier: launching custom skincare lines requires significant capital and manufacturing expertise most don't have.
Private label manufacturing eliminates this obstacle. These partnerships provide turnkey solutions—from FDA-compliant formulation and testing to packaging and fulfillment—that reduce time-to-market and operational complexity.
This guide explores how the right manufacturing partner can help you launch specialized skincare products without the prohibitive upfront investment, focusing on manufacturers who support low minimum orders and custom formulations for emerging brands.
TLDR
- Launch faster with ready-made formulations and FDA-compliant facilities—no infrastructure investment required
- Evaluate manufacturers by certifications (FDA cGMP, ISO), MOQs (500-10,000+ units), and turnaround times (1-12 weeks)
- U.S. manufacturers specialize in clean beauty, cosmeceuticals, natural formulations, and solid formats
- Start with 500 units at entrepreneur-friendly manufacturers or 10,000+ for specialized formats
- Match product quality with scalability, regulatory compliance, and shared brand values
Overview of Private Label Skincare Manufacturing in the USA
Private label skincare manufacturing refers to the practice where manufacturers produce skincare products that brands sell under their own label.
Unlike contract manufacturing (where brands provide custom formulations) or white labeling (selling pre-made products with minimal customization), private label offers a middle ground: stock formulations you can customize with your branding, packaging, and ingredient modifications.
The U.S. private label cosmetics market demonstrates impressive momentum, valued at $10.64 billion in 2024 and projected to reach $14.39 billion by 2030 at a CAGR of 5.2%. Skincare dominates this market, accounting for 41.7% of total revenue.
Why the USA Leads Private Label Manufacturing
This dominance isn't accidental. The United States has become a manufacturing powerhouse, representing 89.24% of North American private label market revenue. Several factors drive this leadership:
- FDA oversight and MoCRA compliance - The Modernization of Cosmetics Regulation Act mandates facility registration and product listing, ensuring rigorous safety standards
- Clean beauty innovation - U.S. manufacturers lead in developing toxin-free, natural, and organic formulations that meet consumer demand
- Advanced R&D capabilities - Domestic facilities invest heavily in research, creating formulations that rival or exceed national brand quality
- Supply chain proximity - Manufacturing in the U.S. reduces shipping times and simplifies compliance with domestic regulations
The Private Label Process
Understanding how these advantages translate into your product launch helps set realistic expectations. The typical private label journey follows these stages:
- Formulation Selection/Development (1-4 weeks) - Choose from stock formulas or request modifications
- Compliance and Testing (2-12 weeks) - Ensure FDA compliance, stability testing, and safety verification
- Packaging Design (2-4 weeks) - Select containers, labels, and branding elements
- Production (1-8 weeks) - Manufacture products according to specifications
- Fulfillment (1-2 weeks) - Package, label, and ship to your warehouse or customers
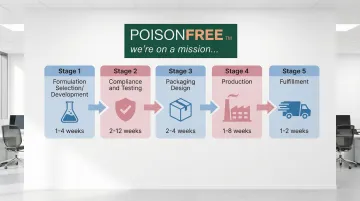
The timeline varies dramatically based on your approach. Stock formulations typically require 4-8 weeks from selection to delivery, while fully custom development extends to 12-24 weeks due to testing requirements.
The USA hosts dozens of private label manufacturers, each with different strengths. Selecting the right partner requires understanding certifications, specializations, minimum order quantities, and how well they align with your brand positioning.
The manufacturers profiled in this guide represent the leading options across various specializations, from clean beauty formulations to high-volume production capabilities.
Top Private Label Skincare Manufacturers in the USA
This list represents manufacturers with proven track records, diverse capabilities, and strong reputations for quality, compliance, and customer service.
We selected manufacturers based on:
- FDA/ISO certifications and compliance standards
- MOQ flexibility for startups and established brands
- Product range and formulation capabilities
- Turnaround times and production speed
- Market positioning across different price points and specializations
Poison-Free Private Label
Poison-Free Private Label is a family-owned manufacturer based in Northern Texas with 30 years of experience, specializing in 100% toxin-free, poison-free, and petrochemical-free formulations. Founded by Sindi Holmlund, who started the company in her kitchen to create safer alternatives for her family, the company operates on a simple philosophy: "If I won't use it, I won't make it and sell it to anyone else."
What sets Poison-Free apart is their entrepreneur-friendly approach:
- Extremely low MOQ of just 500 units—priced as if you bought 10,000
- Orders typically ship within one week once materials are in-house
- Eliminates financial barriers that prevent many startups from launching
The formulation library includes over 100 poison-free products made exclusively from plants, minerals, and vitamins.
For brands seeking clean beauty credentials without compromise, Poison-Free offers formulation duplication services that remove harmful ingredients from existing products and replace them with safe alternatives.
| Criteria | Details |
|---|---|
| Minimum Order Quantity | 500 units (one of the lowest in the industry, ideal for startups and market testing) |
| Turnaround Time | Typically ships within 1 week once materials are in-house |
| Specialization | 100% toxin-free, poison-free, and petrochemical-free personal care and household products |
| Certifications | FDA cGMP compliant manufacturing facility |

Cosmetic Solutions (Florida)
Cosmetic Solutions, founded in 1991 and headquartered in Boca Raton, operates a 100,000 sq. ft. innovation campus focused on luxury, medical-grade, and clean beauty formulations. They serve medical professionals, spas, and established brands seeking high-performance skincare that competes with premium national brands.
Comprehensive turnkey services include R&D, custom formulation development, packaging design, and fulfillment.
Cosmetic Solutions positions itself as a partner for brands that prioritize clinical efficacy and premium positioning, offering OTC (over-the-counter) drug products alongside cosmetics.
| Criteria | Details |
|---|---|
| Minimum Order Quantity | Low MOQs for private label; higher for custom contract manufacturing (typically 1,000+ units) |
| Turnaround Time | 8-12 weeks for custom formulations; faster for stock products |
| Specialization | High-performance skincare, body care, hair care, and OTC products with medical-grade positioning |
| Certifications | FDA Registered, cGMP Compliant, ISO 22716 Certified |
RainShadow Labs (Oregon)
Established in 1983 and celebrating over 40 years in business, RainShadow Labs in St. Helens, Oregon, specializes in natural, organic, and cruelty-free personal care products. They're ideal for indie brands and spas seeking clean formulations backed by sustainable manufacturing practices, including wind-powered facilities.
RainShadow offers exceptional flexibility with their "Buy Direct" program that requires no minimums for wholesale finished goods, alongside traditional private label services. The stock formulation library includes 100+ options covering serums, masks, scrubs, and more. Sample orders ship within one week, allowing rapid product evaluation.
| Criteria | Details |
|---|---|
| Minimum Order Quantity | 10 gallons for stock formulations (approx. 400-6,000 units depending on size); 25 gallons for custom; no minimum for Buy Direct program |
| Turnaround Time | 8-10 weeks after packaging approval; samples ship in 1 week |
| Specialization | Natural, organic, cruelty-free formulations with 100+ stock options; sustainable wind-powered manufacturing |
| Certifications | FDA Registered, ISO 22716 Certified, GMP Certified, Cruelty-Free |
Columbia Cosmetics (California)
Founded in 1978 in the San Francisco Bay Area, Columbia Cosmetics operates a 65,000 sq. ft. facility and ranks among the largest private label manufacturers in North America. They specialize in both color cosmetics and skincare, serving brands from mass-market to prestige positioning.
Columbia's extensive capabilities include custom formulation, a vast stock color cosmetics library, and specialized services for OTC, organic, and CBD formulations. The company's COSMOS/ECOCERT capabilities make them particularly attractive for brands seeking European organic certification alongside U.S. compliance.
| Criteria | Details |
|---|---|
| Minimum Order Quantity | Generally 1,000+ units for custom formulations; varies by product category |
| Turnaround Time | 10-16 weeks for custom formulations; 6-8 weeks for stock products |
| Specialization | Extensive color cosmetics library alongside skincare; OTC, organic, and CBD formulations |
| Certifications | FDA Registered Facility, FDA/CDPH Drug & Cosmetic Manufacturing Licenses, ISO 22716, COSMOS/ECOCERT capabilities |
Twincraft Skincare (Vermont)
Founded in 1972 and headquartered in Winooski, Vermont, Twincraft Skincare specializes in sustainable bar soap and solid skincare formats. The company's "cleaner approach" philosophy emphasizes innovation in solid formats including premium bar soaps, syndet bars, solid shampoos, and deodorants.
Twincraft's specialization in solid formats means higher MOQs due to production scale requirements—10,000 units for stock soap bases and up to 56,000 units (8 metric tons) for custom bar soap formulations. Still, the company's expertise in this niche makes them the go-to partner for brands focusing on zero-waste and sustainable solid skincare.
| Criteria | Details |
|---|---|
| Minimum Order Quantity | 10,000 units for stock soap bases; 8 metric tons for custom bars (approx. 56,000 units); 1,700 kg for deodorants (approx. 20,000 units) |
| Turnaround Time | 12-16 weeks for custom formulations; 8-10 weeks for stock bases |
| Specialization | Premium bar soaps, syndet bars, solid shampoos, deodorants, and liquid skincare with sustainability focus |
| Certifications | ISO 22716, FDA Registered |
How to Choose the Right Private Label Skincare Manufacturer
Verify Certifications and Regulatory Compliance
FDA cGMP (Current Good Manufacturing Practices) compliance isn't optional—it's essential. These regulations ensure manufacturers maintain sanitary facilities, follow quality control procedures, and produce safe, consistent products. Under the Modernization of Cosmetics Regulation Act (MoCRA), the FDA has mandatory recall authority if products cause serious health consequences.
ISO 22716 represents the international standard for cosmetics GMP, covering production, control, storage, and shipment. While voluntary in the U.S., it serves as the benchmark for compliance and demonstrates a manufacturer's commitment to quality management systems.
Non-compliant manufacturing creates these risks:
- Product recalls that damage brand reputation and create financial losses
- FDA Warning Letters and enforcement actions
- Import refusals for products manufactured in non-compliant facilities
- Microbial contamination and safety issues that endanger consumers
Request certification copies and verify facility registration through the FDA database before committing.
Evaluate MOQ Requirements Relative to Your Business Stage
Minimum order quantities directly impact your initial investment and inventory risk. Startups benefit from **low MOQs (under 1,000 units)** that allow market testing without massive capital commitment. MOQs typically range from 100-500 units for white label programs to 10,000+ units for specialized formats.
Match your MOQ to your business stage:
- Market testing phase: 250-500 units allow you to validate demand before scaling
- Initial launch: 1,000-2,000 units provide sufficient inventory for early sales channels
- Growth phase: 5,000+ units unlock better per-unit pricing and economies of scale
Lower MOQs mean higher per-unit costs, so calculate your break-even point and projected sales velocity before committing.
Assess Customization Capabilities
The customization spectrum ranges from simple label application to complete formulation development:
Stock formulas with custom branding - Fastest and most affordable. Choose from existing formulations and apply your label. Ideal for rapid market entry.
Semi-custom modifications - Adjust fragrances, active ingredient concentrations, or add specific botanical extracts to stock bases. Moderate cost increase with differentiation benefits.
Full custom formulation - Develop unique formulas from scratch. Highest cost and longest timeline (12-24 weeks), but offers complete product differentiation and potential IP ownership.
When evaluating manufacturers, verify they can support your specific product claims (anti-aging, brightening, etc.) and provide transparency on ingredient sourcing and active concentration limits.

Evaluate Turnaround Times and Production Capacity
Faster isn't always better if it compromises quality or indicates the manufacturer is overextended. Typical timelines include:
- Stock formulations: 4-8 weeks
- Semi-custom modifications: 8-12 weeks
- Full custom development: 12-24 weeks
Ask about lead times for raw materials, production schedules, and the manufacturer's capacity to scale with your growth. A manufacturer maxing out at 2,000 units becomes a bottleneck as you scale.
Common Mistakes When Selecting a Manufacturer
Prioritizing Price Over Quality and Compliance
Choosing the cheapest manufacturer often backfires. Low prices may indicate shortcuts in ingredient quality, inadequate testing, or non-compliant facilities.
These issues lead to formulation problems, regulatory violations, or inconsistent product quality that damages your brand reputation far more than the initial savings justify.
The cost of a product recall—both financially and reputationally—far exceeds any savings from selecting a budget manufacturer. Prioritize quality, compliance, and reliability over rock-bottom pricing.
Not Clarifying Intellectual Property and Exclusivity Terms
Who owns your formula? This critical question gets overlooked until it's too late. Understanding ownership from the start protects your investment:
- Stock formulations: Manufacturer retains ownership and can sell identical products to competitors
- Custom formulations: May grant you IP ownership, but only if clearly stated in written agreements
- Default scenario: Without clear contracts, manufacturers retain ownership even when you pay for custom development
Protect your formulations by securing these agreements upfront:
- Non-Disclosure Agreements (NDAs) before sharing concepts
- Development agreements specifying IP ownership terms
- Written contracts reviewed by legal counsel

Failing to Request Samples and Conduct Due Diligence
Beyond contracts and IP terms, product quality verification is essential. Never commit to a manufacturer without testing their products firsthand. Request samples of formulations similar to what you plan to order. Evaluate texture, scent, efficacy, and packaging quality against your brand standards.
Due diligence should include:
- Testing product samples for 2-4 weeks
- Visiting facilities if possible to assess cleanliness and operations
- Checking references from current clients
- Verifying certifications through official databases
- Reviewing contracts with legal counsel before signing
The manufacturer's existing product quality directly predicts what you'll receive. Disappointing samples mean disappointing production runs.
Conclusion
Selecting the right private label skincare manufacturer is a strategic decision that impacts product quality, brand reputation, time-to-market, and profitability.
The best choice depends on your specific needs—whether you're a startup requiring low MOQs and fast turnaround, or an established brand seeking custom formulations and scalable production capacity. Your manufacturer should align with your brand values, target market positioning, and growth trajectory.
Before committing to a manufacturer:
- Request samples from multiple companies
- Verify all certifications and compliance standards
- Have detailed conversations about capabilities, timelines, and terms
- Assess their responsiveness and willingness to collaborate
The right partner should be a collaborative ally that supports your vision, not just a transactional vendor.
For entrepreneurs seeking toxin-free formulations, Poison-Free Private Label offers 500-unit minimum orders priced as if you bought 10,000, combined with fast turnaround times and 30 years of clean beauty expertise. Contact them at customersupport@poisonfree.biz to discuss your private label needs.
Frequently Asked Questions
What is the minimum order quantity for private label skincare manufacturers?
MOQs typically range from 12-500 units for entrepreneur-friendly manufacturers to 1,000-5,000+ units for larger operations. Lower MOQs enable market testing with less risk but carry higher per-unit costs, while specialized formats like bar soaps may require 10,000+ units.
How long does it take to launch a private label skincare line?
Timelines range from 4-8 weeks for stock formulations to 12-24 weeks for custom development. Key factors include formulation complexity, stability testing (often 3 months for custom formulas), packaging customization, and production schedules.
What certifications should I look for in a skincare manufacturer?
Look for FDA registration, cGMP compliance, and ISO 22716 certification to ensure quality control, sanitary facilities, and consistent production. Additional certifications like organic (COSMOS/ECOCERT) or cruelty-free may align with your brand positioning.
What's the difference between private label and contract manufacturing?
Private label uses existing formulations with custom branding (manufacturer owns formula), while contract manufacturing creates custom formulas from scratch with potential brand IP ownership. Private label is faster (4-8 weeks) and more affordable, while contract manufacturing offers uniqueness but needs higher MOQs and 12-24 week timelines.
How much does it cost to start a private label skincare brand?
Small startups typically invest $3,000-$10,000 for initial inventory with low-MOQ manufacturers, covering basic packaging and branding. Professional launches with custom formulations and premium packaging require $20,000-$50,000+, including development, testing, and compliance costs.
Can I customize formulations with private label manufacturers?
Most manufacturers offer customization from adjusting fragrances and active ingredients in existing formulas to developing completely custom formulations. Stock modifications maintain lower MOQs, while full custom development requires 1,000-5,000+ units and 12-24 week lead times for R&D and testing.


