
Introduction
Formulation errors in gel toothpaste lead to product separation, inconsistent texture, and failed stability testing—mistakes that can sink a new product line before it launches.
This guide helps entrepreneurs, private label manufacturers, cosmetic formulators, and small businesses master gel toothpaste development through precise ingredient selection, processing techniques, and quality control.
The global tooth gel market is projected to reach $21.1 billion by 2025, growing at 4.5% annually.
For businesses entering this space, understanding the technical specifics of gel formulation determines whether your product succeeds or fails regulatory compliance and shelf-life testing.
This article covers the complete formulation process—from ingredient selection through final product—identifies factors affecting stability and performance, and clarifies when gel formats offer strategic advantages over traditional paste.
TLDR
- Gel toothpaste uses gelling agents, humectants, and active ingredients to create transparent, stable products
- Gel format delivers superior transparency, smoother texture, and better active ingredient suspension
- Proper formulation maintains pH 6.5-7.0 and ensures consistent texture across production batches
- Key challenges: air bubbles, viscosity control, ingredient compatibility, and shelf stability
What Is Gel Toothpaste Formulation?
Gel toothpaste formulation combines gelling polymers, humectants, active ingredients, flavoring agents, and preservatives to create a clear or translucent oral care product. The result is a stable gel that delivers cleaning, whitening, or therapeutic benefits while maintaining the right consistency for dispensing and use.
A gel toothpaste is a water-based semisolid system where a polymeric network holds the liquid phase in place. This creates a structure that doesn't flow under normal conditions.
Gel formulations differ from traditional paste formulations in several ways:
| Feature | Gel Matrix | Paste Matrix |
|---|---|---|
| Primary Structure | Swollen polymeric microgel network (Carbopol) | High solid load (abrasives/fillers) + binders |
| Appearance | Transparent or translucent | Opaque |
| Solids Content | Low (typically 20-30% abrasive) | High (often ~50%) |
| Mechanism | Polymers trap water through swelling | Physical packing of solids + gum thickening |

The key differences come down to structure and appearance:
- Gels rely on water-soluble polymers that create transparency
- Pastes use insoluble abrasives and opacifiers for opacity
- Flow behavior depends on the formulation approach—polymer networks vs. solid particle packing
Why Gel Formulation Is Used in Oral Care Products
Choosing a gel format is a strategic decision driven by both market positioning and functional advantages. Consumer preference for transparent products perceived as "cleaner" has made gels particularly attractive for premium positioning.
The format also allows manufacturers to showcase colored actives or sparkle effects, provides a smoother mouthfeel, and creates clear marketing differentiation from traditional pastes.
Market Demand and Consumer Preferences
The clear gel toothpaste segment is identified as a leading application and key growth driver, specifically due to consumer preference for visually appealing, translucent products. According to Mintel's US Oral Care Market Report, ingredient transparency has become a top priority for American consumers, and the inherent transparency of gel formulations visually reinforces claims of purity.
Key market drivers include:
- Aesthetic appeal: Consumers associate transparency with cleanliness and premium quality
- Premium positioning: Gels typically command higher price points than traditional pastes
- Gentler abrasion profiles: Lower RDA (Relative Dentin Abrasivity) values make gels suitable for sensitive teeth
- Enhanced active delivery: Gel matrices allow certain ingredients to work more effectively
Functional Advantages
Gel matrices offer distinct technical benefits that go beyond visual appeal. Research indicates that Carbopol polymers exhibit bioadhesive properties, helping formulations adhere to teeth and gums.
This prolongs the retention time of active ingredients like fluoride, whitening agents, and desensitizers, potentially enhancing their effectiveness.
Studies show that Carbopol 980, when combined with fluoride and stannous chloride, significantly protects enamel against erosive wear. The polymer network keeps active ingredients in close contact with the tooth surface longer than traditional paste formats.
What Goes Wrong Without Proper Formulation
Without proper gel formulation expertise, manufacturers face several critical failures:
- Phase separation: Water separates from the gel matrix (syneresis)
- Inconsistent texture: Lumpy or grainy mouthfeel
- Poor stability: Product degrades before expiration date
- Inadequate viscosity: Product runs off toothbrush or doesn't dispense properly
These failures aren't just aesthetic: they represent product recalls, customer complaints, and brand damage. For small businesses and entrepreneurs, a single failed batch can mean significant financial loss.

How the Gel Toothpaste Formulation Process Works
Gel toothpaste formulation follows six key stages: ingredient preparation and weighing, hydration of gelling agents, adding active ingredients and additives, pH adjustment, homogenization, and quality testing.
Shortcuts or improper sequencing lead to formulation failure. Each stage builds on the previous one.
Core Ingredients and Their Roles
What goes into the process:
- Gelling polymers: Carbopol 940/934 (0.4-0.9% concentration)
- Humectants: Glycerin, propylene glycol (5-10%)
- Active ingredients: Fluoride, whitening agents, herbal extracts
- Preservatives: Methylparaben, propylparaben
- Sweeteners: Saccharin, xylitol
- Flavoring agents: Essential oils, flavor compounds
- Purified water: Base liquid phase
These ingredients work together through a chemical transformation. Gelling polymers hydrate and swell in water. pH adjustment (usually with triethanolamine) triggers gel network formation, creating a three-dimensional polymer matrix that traps water and other ingredients.
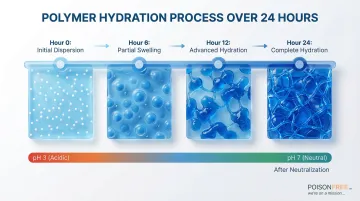
Step 1: Polymer Hydration and Gel Base Preparation
This first step determines final product quality. Disperse the gelling agent (Carbopol at 0.4-0.9% concentration) slowly into water with gentle stirring to prevent clumping.
The polymer needs 12-24 hours to hydrate fully.
Critical parameters:
- Dispersion method: High-shear mixing prevents clump formation
- Temperature: Heating water to 80-85°C ensures full polymer extension
- Hydration time: Rushing this step creates "fish eyes" (visible lumps)
- Neutralization: pH adjustment triggers gel network formation
Carbomer dispersions are acidic (pH ~3). Adding a base neutralizes acid groups, causing the polymer to swell up to 1,000 times its volume and build viscosity.
Incomplete hydration is the most common cause of formulation failure.
Step 2: Addition of Active Ingredients and Additives
Add ingredients sequentially to prevent incompatibilities:
- Preservatives first: Ensure microbial protection throughout mixing
- Humectants: Add glycerin (5-10%) for moisture retention
- Active ingredients: Add herbal extracts, essential oils, or therapeutic agents with continuous mixing
- Air management: Mix to ensure uniform distribution without excess air
Key considerations:
- Carbomers are negatively charged and react with positively charged ingredients, forming precipitates
- High salt levels collapse the gel network by shielding electrical charges
- Add temperature-sensitive ingredients after cooling
- Vacuum mixing at 27 in. Hg prevents air entrapment
Step 3: Homogenization, pH Adjustment, and Quality Control
Final processing ensures consistency and safety. Homogenize the mixture to eliminate remaining lumps or aggregates. Test and adjust pH to the target range of 6.5-7.0, which is safe for the oral cavity.
Quality checkpoints:
- Viscosity verification: Must match target specifications for dispensing
- Appearance: Clarity and color uniformity
- Homogeneity: No separation or settling
- Spreadability: Appropriate texture on toothbrush
- Stability: Sample testing for shelf life prediction
ANSI/ADA Standard 130 requires pH < 10.5, but most formulations target 6.5-7.0 to prevent enamel damage (which occurs at pH ~5.5).
Where Gel Toothpaste Formulation Is Applied
Gel formulation occurs in various manufacturing settings, each with different capabilities and minimum order requirements. Understanding where formulation happens helps entrepreneurs select appropriate manufacturing partners.
Manufacturing Settings
Types of facilities:
- Private label cosmetic manufacturers: Custom formulations for multiple clients
- Contract manufacturing facilities: Large-scale production (higher MOQs)
- In-house production labs: Established brands with exclusive formulations
- Small-batch artisanal producers: Low-volume specialized production
For entrepreneurs testing market entry, finding the right manufacturing partner is crucial. Poison-Free Private Label offers 500-unit minimum orders—ideal for validating product-market fit without heavy upfront investment.
Most facilities require 1,000 to 5,000 units per order, creating barriers for new businesses. Lower MOQs allow start-ups to test products and gather customer feedback before committing to large production runs.
Product Lifecycle Stages
Understanding when formulation happens in your product journey helps you plan resources and timelines. Manufacturers create formulations during these stages:
- Initial product development: Prototype creation and testing
- Scale-up: Lab batch (100-500g) to production batch (50-500kg)
- Reformulation: Product improvement or cost optimization
- Custom formulation: Specific private label requirements
Project Triggers
Common reasons businesses initiate gel formulation projects:
- Market demand for premium oral care products
- Desire to create natural or toxin-free alternatives to conventional toothpastes
- Need for specialized formulations (sensitivity, whitening, herbal)
- Entrepreneur/business entry into oral care market
Initial formulation is typically a one-time development process. However, you may need periodic reformulation due to:
- Ingredient availability changes or supplier issues
- Regulatory updates requiring compliance adjustments
- Stability issues discovered during shelf-life testing
- Product line extensions building on successful formulas
Key Factors That Affect Gel Toothpaste Formulation
Success depends on controlling multiple variables that interact in complex ways. Understanding these factors prevents costly formulation failures.
Ingredient Selection and Compatibility
Gelling agent concentration directly determines viscosity and texture:
- 0.2-3.0% Carbopol concentration range
- 0.35% creates translucent fluoride gel
- 0.50% creates clear fluoride gel
- Extreme pH levels require higher concentrations
Humectants control moisture retention and mouthfeel:
- Sorbitol (70% solution) often used at 68% to match silica refractive index
- Glycerin typically 9-15%
- Balance required to prevent drying or excessive moisture
Your preservative system must remain effective at neutral pH:
- USP <51> requires 1.0 log reduction in bacteria by day 14
- ISO 11930 requires 3 log reduction by day 7
- Anionic polymers can interact with cationic preservatives
Active ingredient stability varies by type:
- Fluoride compounds require specific pH ranges
- Whitening agents may be temperature-sensitive
- Herbal extracts must not separate from gel matrix
Operating Conditions
Once you've selected compatible ingredients, mixing parameters determine how effectively they combine:
- Speed — High-shear mixing creates smooth texture but may introduce air bubbles
- Duration — Insufficient mixing causes poor consistency throughout the batch
- Temperature — Affects ingredient solubility and how quickly the gel forms
- Order of addition — Adding ingredients in the correct sequence prevents incompatibilities
Equipment requirements include:
- High-shear mixers for smoother texture
- Accurate pH meters for proper adjustment
- Filling equipment rated for 70,000-100,000 cP viscosity range
- Contamination-free storage tanks
Scale Considerations
Lab-scale batches (100-500g) behave differently than production batches (50-500kg) due to mixing dynamics. Scaling up requires adjustment of mixing times and sometimes polymer concentrations.
For entrepreneurs testing new formulations, working with manufacturers that offer low minimum order quantities (such as 500 units rather than 10,000+) allows you to validate market demand before committing significant capital to large production runs.
Safety and Regulatory Constraints
Oral care products face specific regulatory requirements:
- pH must be 6.5-7.0 to avoid oral irritation
- All ingredients must be FDA-approved for oral use
- Preservative levels must prevent microbial growth while remaining safe
- Heavy metals testing required
- Fluoride limited to maximum 276 mg per package for dentifrices
Toxin-free formulations require careful ingredient sourcing and verification to ensure all components meet safety standards.

Common Formulation Issues and Misconceptions
Understanding common mistakes prevents costly errors and reformulation cycles.
Incorrect Assumptions
Three misconceptions plague gel toothpaste formulation:
- Excessive gelling agent improves texture — False. The optimal concentration range is narrow (0.4-0.9%). Exceeding this creates sticky, unpleasant feel and degrades sensory properties.
- Gels are inherently more stable than pastes — False. Gels require more careful pH and preservative management since the polymer network is sensitive to electrolytes and pH shifts that don't affect paste formulations.
- Clear appearance indicates chemical-free formulation — False. Polymers create the transparency, not natural ingredients. Clear gels still contain synthetic polymers and preservatives.
Process Oversimplification
Beyond ingredient misconceptions, formulators often oversimplify the manufacturing process itself.
Common formulator errors:
- Skipping proper hydration time leads to lumpy texture and poor clarity
- Adding all ingredients at once causes incompatibilities and precipitation
- Forgetting pH adjustment results in thin or unstable gels
- Using incompatible essential oils without testing causes separation from the gel matrix
Confusion Between Process and Outcome
Initial success doesn't predict long-term performance. Proper viscosity at production doesn't guarantee stability over 24 months, and matching a competitor's texture doesn't mean your formulation will perform identically under all storage conditions.
Testing requirements that can't be skipped:
- Stability testing through full shelf life (typically 24-36 months)
- Temperature cycling to reveal weaknesses invisible at room temperature
- Microbial challenge testing to confirm preservative effectiveness

When Gel Formulation May Not Be Appropriate
Gel format isn't always the right choice. Understanding when paste format is superior helps you make the right choice.
Situations Where Gel Is Unnecessary or Inefficient
High levels of insoluble abrasives required:
Gel matrices have lower tolerance for solids than pastes. While pastes handle ~50% abrasive load, gels typically cap at 20-30%.
For heavy stain removal requiring aggressive abrasion, paste format handles particulates better.
Cost constraints favor simpler formulations:
Specialized high-clarity silica and rheology modifiers (Carbopol) carry premiums over standard calcium carbonate and CMC used in opaque pastes.
Manufacturing costs also run higher due to energy-intensive heating/cooling cycles and vacuum processing.
Target market prefers traditional white paste:
Many consumers still prefer traditional paste appearance and lower price points. The "natural" consumer segment may reject synthetic carbomers.
Constraints That Reduce Effectiveness
Certain active ingredients don't suspend well in gel matrices:
- High levels of calcium compounds (historically incompatible with Carbopol)
- Some whitening agents require paste matrix
- Very long shelf life requirements may be harder to achieve
Very low-cost positioning makes gel's premium ingredients economically unfeasible. The format typically requires premium pricing to justify ingredient and processing costs.
Signals of Poor Strategic Fit
Warning signs gel is chosen by default rather than strategic fit:
- Lack of clear product differentiation beyond appearance
- No consumer research supporting gel preference in target market
- Inability to justify premium pricing that gel format typically commands
- Product concept doesn't leverage gel's functional advantages
When these signals appear, reconsider whether paste format better serves business objectives.
Conclusion
Gel toothpaste formulation creates aesthetically appealing, stable oral care products through systematic combination of gelling polymers, active ingredients, and careful processing that delivers specific texture, appearance, and performance characteristics. The process requires technical precision—from polymer hydration through pH adjustment to final quality testing.
Mastering this technical precision delivers clear advantages for oral care product developers:
- Creates high-quality products that meet consumer expectations
- Ensures regulatory compliance and safety standards
- Prevents costly formulation failures during development
- Enables informed decisions about ingredient selection and manufacturing partnerships
For entrepreneurs and small businesses, partnering with experienced manufacturers who offer low minimum order quantities provides a practical entry point into the growing gel toothpaste market without large upfront investment. Poison-Free Private Label's 500-unit MOQ, for example, allows market testing of new formulations while maintaining the same per-unit pricing as large orders.
Your path forward involves balancing aesthetic appeal with functional performance, understanding regulatory requirements, and selecting manufacturing partners with proven formulation expertise in toxin-free oral care products.
Frequently Asked Questions
What are the ingredients in toothpaste gel?
Gel toothpastes contain gelling agents (Carbopol/carbomer at 0.4-0.9%), humectants (glycerin, propylene glycol), active ingredients (fluoride, whitening agents, or herbal extracts), preservatives, sweeteners, flavoring agents, and purified water. The transparent appearance comes from water-soluble polymers instead of insoluble abrasives.
Does anyone make gel toothpaste?
Major oral care brands produce gel toothpastes, and private label manufacturers like Poison-Free Private Label offer custom formulation services with 500-unit minimums. This enables entrepreneurs to launch their own gel toothpaste brands without large upfront investment.
What's the difference between toothpaste and toothpaste gel?
Gels are transparent and use water-soluble polymers, while pastes are opaque and use insoluble abrasives. Gels have a smoother texture and gentler abrasion levels, though both formats deliver similar cleaning benefits.
