
Understanding Toothpaste Formulation Science
Many entrepreneurs, formulators, and health-conscious consumers don't realize that toothpaste contains 10-15+ carefully balanced ingredients working in concert. Each component serves a specific purpose—from mechanical cleaning to disease prevention to sensory satisfaction.
Yet ingredient transparency remains frustratingly opaque in an industry where 67% of conventional toothpastes contain potentially irritating surfactants like sodium lauryl sulfate.
That's why understanding formulation science matters—whether you're launching a clean-label brand, seeking natural alternatives, or simply want to know what you're putting in your mouth twice daily.
This guide decodes ingredient categories, examines evidence-based natural alternatives, and reveals the formulation principles that separate effective products from marketing hype.
TLDR:
- Toothpaste contains 10-15+ ingredients across six key categories
- Natural toothpaste market projected to reach $4.1 billion by 2031 (9.0% annual growth)
- Nano-hydroxyapatite demonstrates efficacy comparable to fluoride in clinical trials
- RDA values under 250 are considered safe—lower doesn't mean safer
- Ingredient compatibility (like fluoride + calcium carbonate) can destroy product efficacy
Essential Toothpaste Ingredients: The Building Blocks
Abrasives: The Cleaning Powerhouse
Abrasives mechanically remove plaque, debris, and extrinsic stains from tooth surfaces through controlled friction. Their effectiveness depends on particle hardness, size, shape, and concentration.
Understanding RDA (Relative Dentin Abrasivity):
The ISO 11609 standard sets a maximum RDA of 250 as the safety threshold. Any toothpaste at or below this limit is considered safe for lifetime use.
Contrary to marketing claims, clinical studies show no significant difference in dentin wear between products with varying RDA levels below 250. Lower RDA values don't equate to "safer" products but may compromise stain removal effectiveness.
Common Abrasive Types:
- Hydrated Silica: Compatible with all fluoride types; ideal for clear gel formulations due to refractive index matching
- Calcium Carbonate: Insoluble white powder that's incompatible with sodium fluoride—forms insoluble salts that reduce fluoride availability
- Dicalcium Phosphate Dihydrate: Neutral pH with good compatibility; can dehydrate over time, becoming harsher
- Sodium Bicarbonate: Very low abrasivity (approximately 7 RDA); cleans through both chemical and mechanical action

Humectants: Moisture and Texture Control
Beyond providing the abrasive foundation, toothpaste formulations need ingredients that maintain moisture and create the right texture.
Humectants prevent paste from drying out upon air exposure while creating smooth, spreadable texture and facilitating active ingredient dispersion. Common options include:
- Glycerin (vegetable-derived or synthetic)
- Sorbitol (note: high doses can act as osmotic laxative)
- Xylitol (non-fermentable by oral bacteria, offering additional anticaries benefits)
- Propylene glycol and polyethylene glycol
These ingredients typically make up 20-30% of formulation weight, directly influencing mouthfeel and product stability over shelf life.
Binders and Thickeners: Achieving the Perfect Consistency
While humectants maintain moisture, binders create structural integrity. These hydrophilic colloids stabilize formulations by preventing phase separation between solids and liquids. They determine viscosity, influence foaming characteristics, and affect how easily the product rinses.
Natural Options:
- Xanthan gum
- Carrageenan
- Cellulose gum (sodium carboxymethylcellulose)
- Sodium alginate
These require proper hydration and must be compatible with other formulation components, particularly actives like fluoride or essential oils.
Surfactants and Foaming Agents: Creating the Clean Feel
The right consistency matters little without effective cleaning action. Surfactants lower surface tension to disperse ingredients, penetrate plaque biofilm, and create foam that enhances the perception of cleanliness.
Sodium Lauryl Sulfate (SLS) Concerns:
Found in approximately 67% of toothpastes, SLS is a strong denaturing agent that can damage the mucosal mucin layer. Clinical evidence shows that SLS-free dentifrices statistically significantly reduce the number, duration, and pain of recurrent aphthous ulcers (canker sores) in susceptible patients.
Gentler Alternatives:
- Sodium cocoyl glutamate
- Sodium methyl cocoyl taurate
- Decyl glucoside
- Cocamidopropyl betaine
- Sodium coco-sulfate
These milder surfactants differentiate products for sensitive populations without sacrificing cleaning efficacy.
Flavoring and Sweetening Agents: The Sensory Experience
Even the most effective formulation fails if users won't brush regularly. Flavoring agents mask raw ingredient tastes and provide refreshing sensations critical for consumer compliance. Essential oils must be used within safe limits to avoid toxicity or sensitization.
Safety Considerations:
IFRA standards classify toothpaste under Category 6 (products with oral and lip exposure). For example, cinnamon bark oil is restricted to a maximum of 0.23% in this category.
All flavor ingredients must have approved status (GRAS - Generally Recognized as Safe) for products with potential ingestion.
Natural Sweeteners:
- Xylitol: Non-cariogenic and inhibits S. mutans growth
- Stevia: Natural zero-calorie sweetener
- Erythritol: Sugar alcohol that doesn't promote decay
Functional Ingredient Categories: Beyond Basic Cleaning
Anticaries Agents: Cavity Prevention
Fluoride: The Gold Standard
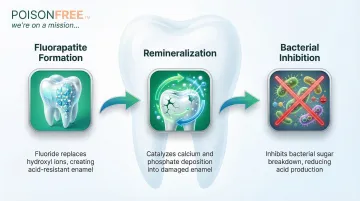
Fluoride remains the most effective known topical substance for caries prevention. It works through three distinct mechanisms:
- Fluorapatite Formation: Fluoride ions replace hydroxyl ions in enamel hydroxyapatite, creating fluorapatite that's more resistant to acid dissolution
- Remineralization: Catalyzes calcium and phosphate deposition into demineralized enamel
- Bacterial Inhibition: Inhibits bacterial glycolysis glycolysis (sugar breakdown), reducing acid production
Fluoride Type Comparison:
| Fluoride Type | Key Properties |
|---|---|
| Sodium Fluoride (NaF) | Releases fluoride ions immediately; requires silica abrasives; incompatible with calcium-based abrasives |
| Stannous Fluoride (SnF2) | Dual action: anticaries + antibacterial; effective against plaque, gingivitis, and sensitivity |
| Sodium Monofluorophosphate (SMFP) | Releases fluoride via enzymatic hydrolysis (enzyme breakdown); compatible with calcium carbonate |
Fluoride-Free Alternatives
Nano-Hydroxyapatite (nHAp): Clinical trials demonstrate nHAp is as effective as fluoride in preventing caries and remineralizing initial lesions. It works by adhering to and filling microscopic enamel defects.
CPP-ACP (Recaldent): Stabilizes calcium and phosphate ions to promote remineralization; clinically proven effective for white spot lesions.
Functionalized Tricalcium Phosphate (fTCP): Designed to enhance fluoride nucleation; clinical trials indicate that fTCP combined with fluoride arrests dentin caries more effectively than fluoride alone.
Xylitol: While non-cariogenic, systematic reviews on its active caries-preventive effect in toothpaste show contradictory results with high risk of bias.
Antimicrobial and Antigingivitis Agents
Natural Antimicrobials:
Research demonstrates that natural agents like neem, green tea, and propolis exhibit comparable effectiveness to chlorhexidine and fluoride in reducing plaque and gingival inflammation.
These alternatives often show fewer side effects (staining, taste alteration) compared to chlorhexidine, potentially improving long-term compliance.
Therapeutic Options:
- Stannous fluoride (dual anticaries/antimicrobial action)
- Zinc compounds (antimicrobial + tartar control)
- Essential oils (tea tree, thyme)
Cosmetic claims (freshening) differ from therapeutic antigingivitis claims, which trigger FDA OTC drug regulations.
Desensitizing Agents for Sensitive Teeth
Desensitizing ingredients work through two mechanisms: blocking dentinal tubules or calming nerve response.
Comparison of Technologies:
- Potassium Nitrate: Depolarizes (calms) nerve endings, preventing pain signal transmission; requires prolonged use (weeks) for significant relief
- Arginine + Calcium Carbonate: Physically occludes (blocks) dentinal tubules by forming a calcium-rich layer; provides both immediate and long-term relief
- Hydroxyapatite: Occludes dentinal tubules to reduce fluid movement and sensitivity
- Stannous Fluoride: Dual mechanism—occludes tubules and provides fluoride benefits
Whitening and Stain-Removal Ingredients
Mechanical vs. Chemical Whitening:
Most natural toothpastes achieve whitening through mechanical abrasion (enhanced silica, baking soda) that removes extrinsic stains. Chemical whitening with peroxides (hydrogen peroxide, carbamide peroxide) targets intrinsic stains but is less common in natural formulations.
Sodium Hexametaphosphate: Prevents stain adhesion by chelating (binding) calcium ions on tooth surfaces.
Natural Approaches:
- Activated charcoal (efficacy debated)
- Baking soda (proven stain removal)
- Enzyme-based systems (papain, bromelain)
Set realistic expectations—natural whitening primarily removes surface stains rather than changing intrinsic tooth color.
Tartar Control and Anti-Calculus Agents
Tartar (calculus) forms when plaque mineralizes. Anti-calculus agents like pyrophosphates and zinc citrate inhibit crystallization (hardening) by interfering with calcium phosphate precipitation.
These agents prevent tartar formation but cannot remove existing calculus—professional cleaning remains necessary.
Natural vs. Synthetic Toothpaste Formulation: A Balanced Perspective
The Rise of Clean Label and Natural Oral Care
The global natural toothpaste market is valued at $2.25 billion in 2024 and projected to reach $4.1 billion by 2031, growing at 9.0% annually. North America dominates with over 40% market share.
Consumer concerns driving this growth:
- SLS and oral irritation
- Triclosan (now largely phased out)
- Artificial sweeteners and synthetic dyes
- Parabens and preservatives
- Microplastics in formulations
The FDA has no formal definition for "natural" in cosmetic labeling. Claims must not be false or misleading, but the term remains largely unregulated, requiring manufacturer integrity and transparency.
Understanding these market forces helps explain why formulators must balance consumer expectations with scientific efficacy.
Natural vs. Synthetic: A Formulation Comparison
| Aspect | Natural Ingredients | Synthetic Ingredients |
|---|---|---|
| Safety Profile | Perceived as gentler; lower mucosal irritation | Extensive safety data; proven efficacy (e.g., fluoride) |
| Quality Consistency | Batch-to-batch variability in plant extracts | Consistent quality and performance |
| Environmental Impact | Biodegradable; no microplastics | Some compounds persist in environment |
| Shelf Stability | Shorter without synthetic preservatives | Extended stability |
| Clinical Evidence | Less research for some ingredients | Decades of clinical validation |
| Formulation Complexity | Requires specialized expertise | Well-established formulation protocols |
| Cost | Higher raw material costs | Cost-effective at scale |
| Consumer Appeal | Aligns with clean label values | May trigger allergen concerns in sensitive users |

Formulating Effective Natural Toothpaste: The Challenge
Replacing synthetic ingredients while maintaining efficacy, stability, and consumer acceptance requires deep formulation expertise. Critical challenges include:
- Ingredient compatibility - pH sensitivity of natural preservatives, essential oil interactions, and natural binder hydration requirements
- Microbial stability - Water-based natural formulas prone to contamination requiring robust preservation efficacy testing (PET)
- Natural preservation systems - Organic acids and benzyl alcohol must remain effective at product pH
- Consumer expectations - Meeting texture, foaming, and flavor preferences without synthetic agents
- Shelf consistency - Maintaining appearance and performance throughout product life
The Poison-Free Approach to Clean Formulation
Poison-Free Private Label operates on a simple philosophy: "if I won't use it, I won't make it and sell it to anyone else."
Founded 30 years ago in Northern Texas, the company has formulated over 100 toxin-free products using only plants, minerals, and vitamins.
What sets this approach apart:
- FDA cGMP compliant manufacturing facility
- Commitment to efficacy alongside safety
- Low minimum order quantities (500 units) for entrepreneurs
- Large raw material inventory for custom formulations
- 30 years of natural formulation expertise
This approach demonstrates that effective natural oral care is achievable when formulators prioritize both safety and performance rather than compromising one for the other.
Regulatory and Testing Considerations for Toothpaste
In the U.S., toothpaste classification depends on claims:
Cosmetic (Freshening, Cleaning, Whitening via Stain Removal):
- Regulated under cosmetic guidelines
- No pre-market approval required
- Must follow labeling and safety regulations
OTC Drug (Anticaries, Antigingivitis, Sensitivity Relief):
- Must comply with FDA Monograph M021 for anticaries products
- Requires "Drug Facts" panel
- Must meet specific testing requirements
If you're making anticaries claims, you'll need to complete specific testing:
- Animal caries reduction studies (or validated alternatives)
- Enamel Solubility Reduction (ESR) OR Fluoride Enamel Uptake (FU)
- Stability testing for total fluoride, available fluoride ion, and pH
The FDA recognizes intraoral appliance (IOA) models using human enamel chips as alternatives to animal testing.
Manufacturers can petition for alternatives by demonstrating equivalent accuracy.
Beyond FDA requirements, the FTC mandates competent and reliable scientific evidence for all health and efficacy claims—even for products marketed as "natural." This applies whether you're manufacturing in an FDA cGMP compliant facility or operating under different standards.
Formulation Challenges and Considerations
Critical Ingredient Compatibility Issues
Fluoride Inactivation:
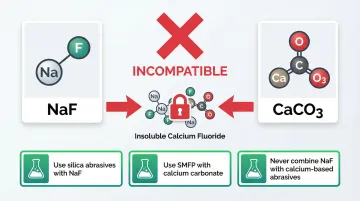
The most common formulation failure occurs when sodium fluoride pairs with calcium carbonate. This pairing creates a critical problem.
Calcium ions react with fluoride to form insoluble calcium fluoride, drastically reducing the fluoride available for cavity protection and rendering the anticaries agent ineffective.
Solutions:
- Use silica abrasives with sodium fluoride
- Use sodium monofluorophosphate with calcium carbonate
- Never combine NaF with calcium-based abrasives
Stannous Fluoride Stability:
SnF2 oxidizes and hydrolyzes in water. Modern formulations use stabilization systems (low water content, chelating agents like gluconate or pyrophosphate) to maintain efficacy and prevent staining.
Preservation Strategies
Natural formulations face significant microbial stability challenges without traditional preservatives like parabens.
For manufacturers committed to toxin-free formulations, effective preservation strategies include:
- Organic acid systems (sorbic acid, benzoic acid)
- Essential oils with antimicrobial properties
- Benzyl alcohol
- Low water activity formulations
- Rigorous challenge testing to validate preservation
Companies like Poison-Free Private Label, which exclusively use plant-based, mineral, and vitamin ingredients, rely on these natural preservation systems combined with thorough microbiological testing to ensure product safety and shelf life.
Texture, Flavor, and Mouthfeel Optimization
Consumer sensory experience determines product success. SLS provides high foam, which traditionally signals "cleaning" to consumers. Yet the growing SLS-free market shows consumers increasingly accept lower-foaming alternatives in exchange for perceived safety benefits.
Balancing Act:
- Sufficient abrasivity for cleaning without harshness
- Appropriate viscosity for dispensing and spreading
- Flavor intensity that masks ingredients without overwhelming
- Foam level that signals "cleaning" to consumers
Flavoring often represents the most expensive formulation component and remains crucial for consumer preference and compliance.
Choosing the Right Formulation Partner for Your Brand
Your choice of contract manufacturer directly impacts product quality, launch timeline, and brand reputation. Focus on these key factors:
Experience with Natural Ingredients:
- Track record formulating with plant-based and mineral ingredients
- Understanding of natural ingredient interactions and stability
- Ability to source high-quality, consistent natural raw materials
Regulatory Knowledge:
- FDA cGMP compliance as baseline
- Understanding of cosmetic vs. OTC drug regulations
- Experience with therapeutic claim substantiation
- Transparent testing protocols
Beyond compliance, consider the practical realities of launching your brand. Many manufacturers require 5,000-10,000 unit minimums, creating barriers for startups testing new products.
Entrepreneur-Friendly Terms:
- Low minimum order quantities (500-1,000 units)
- Competitive pricing that doesn't penalize smaller batches
- Flexibility for formulation adjustments between runs
- Fast turnaround times for time-sensitive launches
Values Alignment:
Your manufacturer's ingredient philosophy should mirror your brand values. If you're building a toxin-free brand, partnering with manufacturers who maintain strict standards—such as excluding petrochemicals, synthetic preservatives, or questionable additives—protects your brand integrity from formulation through final production.
Raw Material Inventory:
Manufacturers maintaining large raw material inventories can:
- Reduce lead times significantly
- Offer greater formulation flexibility
- Accommodate custom requests more easily
- Ship finished orders within days rather than weeks
Frequently Asked Questions
What is the formulation of toothpaste?
Toothpaste blends 10-15+ ingredients including abrasives (mechanical cleaning), humectants (moisture retention), binders (consistency), surfactants (foaming), and flavoring agents. Active ingredients like fluoride or natural alternatives clean teeth, prevent disease, and deliver sensory satisfaction.
What do holistic dentists use instead of fluoride?
Holistic dentists often recommend nano-hydroxyapatite, xylitol, calcium phosphate compounds (CPP-ACP), and remineralizing herbal extracts. Clinical evidence varies, though nano-hydroxyapatite shows the strongest efficacy comparable to fluoride in preventing cavities.
What's the difference between gel and paste toothpaste formulations?
Pastes contain opacifying agents and typically higher abrasive content, creating an opaque appearance and stronger mechanical cleaning action. Gels are translucent, often use lower abrasives and different thickening systems, and may use alternative cleaning mechanisms.
How do I ensure my natural toothpaste formulation is stable and effective?
Work with experienced formulators who understand natural ingredient interactions, conduct stability testing (temperature cycling, real-time aging), and test effectiveness through appropriate methods. Manufacturers like Poison-Free Private Label with FDA cGMP compliance and 30 years of toxin-free formulation experience can ensure both stability and performance.
Can natural toothpaste be as effective as conventional fluoride toothpaste?
Some natural ingredients like nano-hydroxyapatite show promise comparable to fluoride in clinical studies, though fluoride has the most extensive evidence. Well-formulated natural toothpastes can be highly effective when combining multiple remineralizing and antimicrobial agents at proper concentrations.
What certifications should I look for in a toothpaste manufacturer?
FDA cGMP compliance is the baseline requirement. Additionally, look for transparency regarding ingredient sourcing, third-party testing protocols, quality control documentation, and willingness to provide certificates of analysis. For therapeutic claims, ensure the manufacturer understands OTC drug monograph requirements and can support regulatory compliance.
