
Introduction: Navigating Your Manufacturing Options
Entrepreneurs face a critical decision when bringing a product to market: build manufacturing infrastructure or partner with an established manufacturer? This choice impacts startup costs, speed-to-market, and long-term scalability.
Confusion between contract manufacturing, private label, OEM, and other models leads to costly mistakes. Choose the wrong path, and you risk outdated inventory, regulatory violations, or inability to scale.
Between 2011 and 2023, there were 334 cosmetic recalls affecting over 77 million units, with 76.8% caused by microbial contamination. Manufacturing partner selection directly impacts product safety and business viability.
To help you make informed decisions, this guide clarifies the key differences between manufacturing models, provides decision frameworks for selecting the right approach, and explains how to evaluate partners based on compliance, capabilities, and business fit.
TLDR:
- Contract manufacturing gives you exclusive formulations and complete control; private label offers speed and lower investment
- MOQs vary dramatically: 10,000+ units for custom contract manufacturing vs. 500-1,000 for private label
- Private label cuts launch time by 60%+ (4-8 weeks vs. 12-24 weeks)
- FDA cGMP compliance and ISO 22716 certification are non-negotiable for safety
- Choose based on priorities: speed/capital (private label) or differentiation/IP (contract manufacturing)
What is Contract Manufacturing? A Complete Overview
Contract manufacturing is a business arrangement where one company hires another manufacturer to produce goods according to the hiring company's specifications and designs. The brand retains ownership of the formulation and intellectual property while the manufacturer provides facilities, equipment, labor, and expertise.
How the Process Works
Understanding the production timeline helps you plan your product launch effectively.
The contract manufacturing journey begins with formulation development. The hiring company works with the manufacturer to create custom product specifications, including ingredient lists, concentrations, textures, and performance characteristics.
This R&D phase typically takes 8-12 weeks and involves repeated testing to perfect the formula. Once finalized, stability and microbiological testing begins—a mandatory 12-16 week process to ensure product safety and shelf life.
Production setup then includes equipment calibration, raw material procurement, and quality control protocols. Manufacturing runs produce the finished goods according to agreed specifications.

What You Control vs. What the Manufacturer Provides
This division of responsibilities defines the partnership structure.
Your responsibilities as the hiring company:
- Product formulation and specifications
- Ingredient selection and quality standards
- Branding, packaging design, and labeling
- Intellectual property ownership
- Quality acceptance criteria
- Marketing and distribution strategy
What the manufacturer handles:
- Production facilities and equipment
- Manufacturing labor and expertise
- Raw material sourcing and procurement
- Quality control testing and validation
- Regulatory compliance infrastructure
- Logistics management and supply chain coordination
Real-World Examples Across Industries
Electronics: Major smartphone brands use contract manufacturers like Foxconn to produce devices. The brand designs product specifications, while the manufacturer handles assembly, component sourcing, and quality testing.
Cosmetics: Leading makeup brands like Rare Beauty and Fenty leverage contract manufacturers such as Mana Products for custom formulations. These brands create proprietary color cosmetics formulas and rely on manufacturers for specialized production capabilities and regulatory expertise.
Food & Beverage: Supplement companies develop unique vitamin blends with specific ingredient combinations, then partner with contract manufacturers for encapsulation, bottling, and FDA compliance management. This allows brands to launch products without investing in their own production facilities.
What is Private Label Manufacturing? Understanding the Model
Private label manufacturing is a model where manufacturers produce pre-formulated products that retailers or businesses can brand and sell as their own. Unlike contract manufacturing, the manufacturer owns the formulation, and buyers select from an existing product catalog.
The Development Process Difference
Private label cuts out the formulation development phase entirely. Manufacturers maintain libraries of tested, proven formulations across product categories.
Buyers review catalogs, select products that fit their brand positioning, and customize primarily through packaging and branding rather than formula modification.
Because formulas are pre-developed, products have already completed:
- Stability testing and microbiological validation
- Regulatory review and compliance documentation
- Production process refinement
- Supplier relationship establishment
- Quality control procedure validation
All of this happens before you place your first order.
Speed-to-Market Advantage
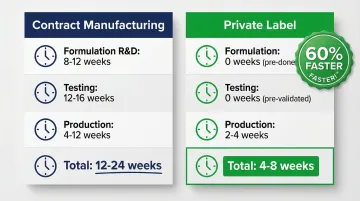
Private label offers a decisive speed advantage, with typical lead times of 4-8 weeks compared to 12-24 weeks for custom contract manufacturing. A 60%+ faster timeline lets brands test market demand with pre-validated formulations before committing to lengthy R&D.
For entrepreneurs prioritizing rapid market entry, the faster launch lets you gather customer feedback, and iterate on packaging or product selection while contract manufacturing projects are still in formulation development.
The Customization Spectrum
Private label customization typically includes:
- Container selection, label design, and color schemes for packaging
- Choosing from available fragrance options
- Adding specific colorants or adjusting fragrance intensity
- Logo placement, product naming, and marketing claims
What remains standardized:
- Base formulation and active ingredients
- Core product performance characteristics
- Manufacturing processes and quality specifications
- Regulatory compliance documentation
Industry Growth and Success Examples
The global private label cosmetics market reached $10.64 billion in 2024, with a projected 5.2% CAGR through 2030. The expansion shows retailers and independent brands leveraging private label for accessibility and speed.
Walmart launched Pretty Smart, a line of 100+ makeup and skincare products priced under $10, emphasizing accessibility. Target and other major retailers use private label partners to create exclusive store brands that offer value and fill portfolio gaps.
Contract Manufacturing vs Private Label: Key Differences Explained
Comparing Product Development and Ownership
Intellectual Property Ownership:
Contract manufacturing grants you complete ownership of the formulation. You control the recipe, and the manufacturer cannot sell copies to competitors. This exclusivity allows for genuine product differentiation and protects your competitive advantage.
Private label means the manufacturer retains formulation ownership. You essentially license the formula. If you switch manufacturers, you cannot take the formulation with you, and the manufacturer may sell the same formula to other brands.
Development Timeline Comparison:
| Stage | Contract Manufacturing | Private Label |
|---|---|---|
| Formulation R&D | 8-12 weeks | 0 weeks (pre-formulated) |
| Testing (Stability/Micro) | 12-16 weeks | 0 weeks (pre-validated) |
| Production Lead Time | 4-12 weeks | 2-4 weeks |
| Total Time-to-Market | 12-24 weeks | 4-8 weeks |
Investment Requirements and MOQs
Minimum Order Quantities:
Contract manufacturing typically requires 10,000 to 50,000 units per SKU to cover setup costs and R&D investment. These high minimums create significant barriers for startups and small businesses testing new products.
Private label offers accessibility, with MOQs as low as 100-500 units for specific categories like serums. Most manufacturers require 1,000 units for better pricing.
Some manufacturers take this further. Poison-Free Private Label, for example, offers 500-unit minimums with pricing calculated as if you bought 10,000 units—removing the traditional volume penalty for small orders.
Upfront Investment Costs:
Beyond order quantities, contract manufacturing demands substantial upfront capital:
- Formulation R&D: $5,000-$25,000
- Stability and safety testing: $3,000-$10,000
- Custom tooling and equipment setup: $2,000-$15,000
- Initial production run: Based on MOQ requirements
Private label eliminates these R&D expenses:
- No R&D or testing expenses (already completed)
- Minimal setup fees (primarily label design)
- Initial investment primarily inventory and branding
- Total starting investment often under $5,000
Control and Customization Trade-Offs
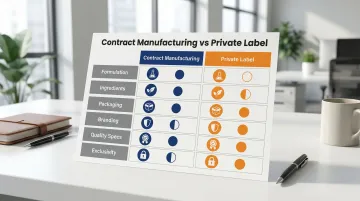
What You Control in Each Model:
| Aspect | Contract Manufacturing | Private Label |
|---|---|---|
| Formulation | Complete control | No control (select from catalog) |
| Ingredients | Select specific sources/grades | Manufacturer's standard suppliers |
| Packaging | Full customization | Select from available options |
| Branding | Complete control | Complete control |
| Quality Specs | Define acceptance criteria | Manufacturer's standards |
| Exclusivity | Exclusive to your brand | Non-exclusive (shared formula) |
The Trade-Off:
Contract manufacturing offers maximum customization and differentiation but requires higher investment and longer timelines. Private label delivers speed and capital efficiency. The limitation? Less product uniqueness. The choice depends on whether your competitive advantage relies on proprietary formulations or on branding, marketing, and customer relationships.
When to Choose Contract Manufacturing vs Private Label
Ideal Scenarios for Contract Manufacturing
Contract manufacturing makes sense when you need full control over formulation. Consider this approach if you:
- Developed unique recipes or ingredient combinations that differentiate your brand and prevent competitors from copying your products
- Operate in crowded markets where performance claims and ingredient innovation drive purchasing decisions
- Need specific ingredient sources, concentrations, or certifications unavailable in standard offerings—like organic certifications or allergen-free formulations
- Project consistent orders of 10,000+ units, where contract manufacturing's unit economics become favorable
The global personal care contract manufacturing market is projected to reach $45.44 billion by 2033 (8.4% CAGR), reflecting brands' shift toward asset-light models with custom products.
Ideal Scenarios for Private Label
Private label works best when speed and capital efficiency matter most. This approach fits when:
- You need to launch within 4-8 weeks to take advantage of trends, seasonal opportunities, or competitive gaps—pre-validated formulations eliminate months of development time
- You're testing market demand before committing to custom development (low MOQs of 500-1,000 units minimize inventory risk)
- You're working with limited capital—startups benefit from eliminating R&D costs, testing fees, and high MOQ requirements (initial investments often start under $5,000 vs. $20,000-$50,000 for contract manufacturing)
- You're entering established categories where differentiation comes from branding and marketing rather than formulation innovation
| Factor | Contract Manufacturing | Private Label |
|---|---|---|
| Time to Market | 3-6 months | 4-8 weeks |
| Initial Investment | $20,000-$50,000 | Under $5,000 |
| Minimum Order Quantity | 10,000+ units | 500-1,000 units |
| Formulation Control | Full customization | Pre-made formulas |
| Best For | Established brands with proprietary needs | Startups testing market demand |

The Hybrid Approach
Many successful brands start with private label for market validation, then transition to contract manufacturing as they scale. This strategy allows you to:
- Launch with private label to validate customer interest and gather feedback quickly
- Generate revenue and establish distribution before investing in custom development
- Use customer feedback to inform custom formulation specifications, then move to contract manufacturing once volume justifies the investment
This approach minimizes risk while preserving the option to develop proprietary formulations as your business matures.
How to Choose the Right Manufacturing Partner: Essential Criteria
Regulatory Compliance Requirements
FDA cGMP Compliance:
Good Manufacturing Practices (GMP) compliance is non-negotiable. The Modernization of Cosmetics Regulation Act (MoCRA) now requires facility registration.
This act grants the FDA mandatory recall authority for adulterated or misbranded products posing serious health risks.
Your manufacturer should maintain FDA cGMP compliance through facility inspections and documented quality systems.
Between 2011 and 2023, microbial contamination caused 76.8% of cosmetic recalls—preventable when manufacturers follow proper GMP protocols.
ISO 22716 Certification:
ISO 22716 is the international standard for GMP in cosmetics, covering production, control, storage, and shipment. This certification demonstrates the manufacturer's commitment to quality beyond minimum regulatory requirements.
Organic and Specialty Certifications:
For organic claims, verify USDA Organic certification (requires 95%+ organic agricultural ingredients) or private standards like COSMOS ORGANIC or Ecocert.
All these certifications require third-party audits and ongoing compliance verification.
Evaluating Manufacturing Capabilities
Production Capacity and Scalability:
Can the manufacturer handle your current needs and future growth? Key questions include:
- Minimum and maximum batch sizes
- Lead times at different volume levels
- Equipment flexibility for various product types
- Capacity to add SKUs as your line expands
Quality Control Processes:
A robust QC system includes:
- Raw material testing to verify identity and purity of incoming ingredients
- In-process controls that monitor production in real-time and catch deviations early
- Finished product testing covering physical, chemical, and microbiological analysis before release
- Complete batch documentation providing traceability from raw materials to finished goods

Supply Chain Reliability:
Strong manufacturers maintain Supplier Qualification Programs and conduct ongoing audits to reduce raw material variability.
Ask about backup suppliers, inventory management systems, and their approach to handling supply disruptions.
Assessing Minimum Order Quantities
Industry standard MOQs for contract manufacturing range from 10,000 to 50,000 units per SKU, justified by setup costs like equipment cleaning and line calibration. However, some manufacturers offer entrepreneur-friendly alternatives.
Finding the Right Fit:
Startups and small businesses should seek manufacturers offering 500-1,000 unit minimums.
Poison-Free Private Label offers 500-unit MOQs with pricing calculated at the 10,000-unit rate—eliminating the typical volume penalty that forces entrepreneurs to overinvest in untested products.
MOQ Complexity Drivers:
Product type affects minimum quantities:
- Serums/Oils: Simpler filling processes often allow 100-250 unit minimums
- Creams/Lotions: Emulsification and heating/cooling cycles typically require 500-1,000 units
- Sunscreen (OTC): Drug regulations and validation requirements push minimums to 2,500-5,000+ units
Communication and Partnership Approach
Responsiveness and Turnaround Times:
How quickly do manufacturers respond to inquiries, provide quotes, and address concerns? Timelines vary dramatically.
Some manufacturers ship orders within a week once materials arrive, while others require 4-12 weeks.
Win-Win Partnership Philosophy:
The best manufacturers view relationships as partnerships, not transactions. Look for manufacturers who:
- Offer guidance on formulation improvements
- Adjust pricing as your volume grows
- Provide flexibility during demand fluctuations
- Proactively communicate about supply chain issues or regulatory changes
This partnership approach has guided Poison-Free Private Label since its founding—where success means helping entrepreneurs launch and scale their brands alongside growing together as the business relationship matures.
Cost Considerations and ROI Analysis
Cost Components Breakdown
Per-Unit Manufacturing Costs:
Unit costs vary significantly by volume. Small-batch production (10,000-50,000 units) carries the highest per-unit costs due to fixed setup fees and lack of bulk ingredient discounts.
Packaging typically represents 5-15% of total product cost.
Setup and Tooling Fees:
Contract manufacturing requires upfront investment:
- Equipment setup and calibration: $1,000-$5,000
- Custom molds or tooling: $2,000-$15,000
- Label plate creation: $200-$500
- Initial formulation development: $5,000-$25,000
Private label eliminates most setup fees, though custom label design may cost $200-$1,000.
Packaging Costs:
Container selection significantly impacts total cost. Premium glass bottles cost $1-$3 each, while standard plastic containers run $0.20-$0.75. Labels, pumps, and secondary packaging add $0.50-$2 per unit.
Shipping and Storage:
Freight costs from manufacturer to your warehouse or fulfillment center vary by distance and volume. If you can't immediately sell or distribute inventory, storage fees apply—typically $50-$200 per pallet monthly.
Understanding these cost components sets the foundation for calculating your actual profitability and return on investment.
Break-Even and ROI Framework
Calculating Break-Even Points:
Break-even analysis must account for all costs:
- Total Investment: MOQ × Unit Cost + Setup Fees + Packaging + Shipping
- Retail Price: Based on market research and competitive positioning
- Variable Costs: Per-unit costs that scale with volume (manufacturing, packaging, shipping)
- Fixed Costs: One-time expenses (setup, tooling, initial inventory)
Example Calculation:
For a moisturizer with $45 retail price:
| Cost Component | Amount |
|---|---|
| Manufacturing cost | $8 per unit |
| Packaging | $2 per unit |
| Total COGS | $10 per unit |
| Gross margin | 78% ($35 profit per unit) |
| MOQ investment | 1,000 units × $10 = $10,000 |
| Break-even point | 286 units (at wholesale pricing) |
Contribution Margin Analysis:
Understanding your contribution margin—the difference between revenue and variable costs—reveals your true profit potential. According to research from Brand Capital Fund, a 10% price increase can transform your economics. Raising that $45 moisturizer to $50 adds $5 contribution margin per unit, potentially generating an additional $5,000 profit on a 1,000-unit run.
Hidden Costs to Watch For
Rush Fees:
Expedited production adds 15-30% to manufacturing costs. Plan ahead. These surcharges eat into margins quickly.
Reformulation Charges:
Changes to approved formulations after initial development incur fees of $500-$2,000 per modification, plus additional testing costs.
Minimum Annual Commitments:
Some manufacturers require minimum annual purchase volumes. Failing to meet commitments may trigger penalties or price increases.
Storage and Warehousing:
If the manufacturer stores finished goods or raw materials on your behalf, expect monthly fees of $50-$300 depending on volume and facility.
Pricing Structures and Volume Thresholds
Costs typically decrease at specific volume thresholds:
- 500-2,500 units: Highest per-unit costs (small-batch pricing)
- 2,500-10,000 units: First price break (15-25% reduction)
- 10,000-50,000 units: Second tier (additional 10-20% reduction)
- 50,000+ units: Bulk pricing (lowest per-unit costs)
These traditional pricing tiers create barriers for entrepreneurs testing new products. Poison-Free Private Label's model addresses this challenge by offering 10,000-unit pricing at just 500 units, eliminating the typical small-batch penalty and allowing entrepreneurs to test products without sacrificing margins.
Frequently Asked Questions
What does contract manufacturing mean?
Contract manufacturing is when one company produces products for another according to their specifications. The hiring company retains ownership of the formulation and intellectual property while the manufacturer provides facilities, equipment, and production expertise.
What is an example of contract manufacturing?
A cosmetics brand hiring a manufacturer to produce their proprietary skincare formula is classic contract manufacturing. The brand owns the unique formulation, while the manufacturer handles production, quality testing, and packaging.
What is the difference between OEM and contract manufacturing?
OEM (Original Equipment Manufacturer) and contract manufacturer are often used interchangeably, both referring to a facility that produces products to the buyer's specifications. The key difference: OEM emphasizes the buyer provides the design, while ODM (Original Design Manufacturer) means the manufacturer owns the design—essentially private label.
What is a manufacturing contract?
A manufacturing contract is the legal agreement outlining product specifications, quality standards, pricing, timelines, intellectual property ownership, and liability provisions. It protects both parties by defining responsibilities and what happens if quality or delivery issues arise.
What are the typical minimum order quantities for contract manufacturing?
MOQs vary widely by industry, typically ranging from 500 to 50,000 units per SKU. Standard contract manufacturing often requires 10,000-50,000 units to justify setup costs, though some manufacturers offer entrepreneur-friendly minimums of 500-1,000 units for startups and small businesses.
How long does it take to launch a product with a contract manufacturer?
Contract manufacturing typically requires 12-24 weeks total (formulation development, testing, and production). Private label dramatically reduces this to 4-8 weeks since formulation and testing are already complete, allowing immediate production once you finalize packaging.


